📌 Key Takeaways
Pre-qualification questionnaires capture capability at a single moment, but consistent supplier performance demands a continuous verification system that connects initial requirements to ongoing monitoring and corrective action.
- Method-Identified Testing Makes Data Comparable: Pairing every reported property with its exact test standard—ISO 287 for moisture, ISO 2758 for burst strength—transforms vague claims into verifiable, audit-ready evidence that can be consistently validated across shipments and suppliers.
- Moisture Measurement Anchors All Other Properties: Because moisture content directly affects burst strength, tensile performance, and dimensional stability, verifying moisture control through proper ISO 287 procedures must come first—inaccurate moisture readings cascade into unreliable data across every downstream mechanical test.
- Risk-Tiering Allocates Verification Resources Efficiently: Remote audits work for established suppliers with documented controls and stable performance, while on-site verification becomes essential for new relationships, repeat defects, or significant process changes—matching audit intensity to actual risk prevents both wasteful over-inspection and dangerous under-verification.
- Four-Part CAPA Closes the Performance Loop: Effective corrective action follows a strict sequence—contain the immediate problem, correct affected material, verify the root cause fix through repeat testing with the same methods, then prevent recurrence by updating systems and controls—with documented evidence at every stage proving the correction actually holds.
- Decision Gates Transform Documentation Into Action: Defining specific triggers—repeat defects within 90 days, calibration drift, documentation inconsistencies, threshold violations—creates automatic escalation rules that catch small problems before they become systemic failures.
Prepared suppliers with method-consistent testing and documented controls earn reduced oversight; gaps trigger immediate corrective protocols.
Procurement managers, quality assurance teams, and supplier compliance leads will find a complete operational framework here, preparing them for the detailed implementation guidance that follows.
Most procurement teams treat supplier pre-qualification as a hurdle to clear once—a box to tick before orders flow. The supplier submits certificates, passes a documentary review, and enters the approved vendor list. Then reality hits: inconsistent grammage, moisture complaints, burst strength failures that disrupt production lines.
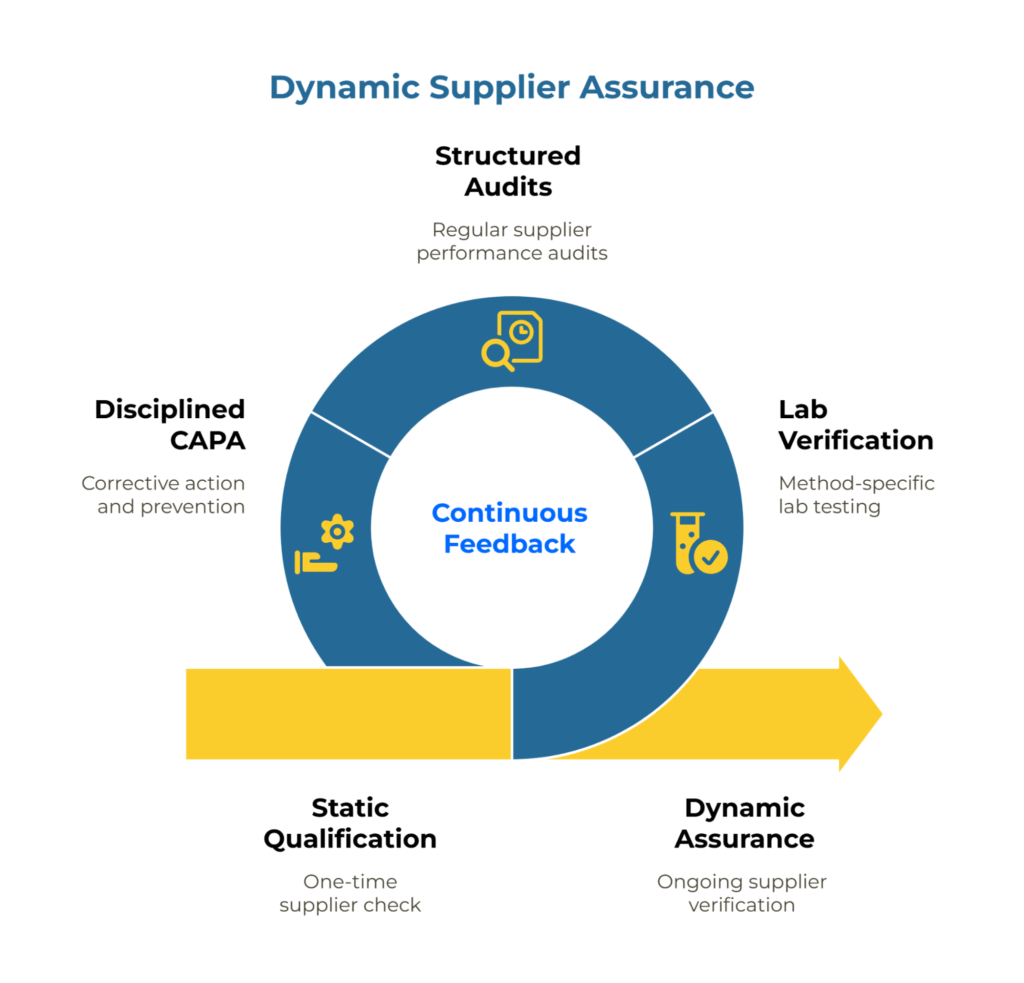
The gap between a clean PQQ (pre-qualification questionnaire) submission and consistent run-rate performance reveals a fundamental flaw in how many organizations approach supplier assurance. Pre-qualification captures a snapshot. What kraft paper buyers actually need is a continuous feedback system that links initial requirements to ongoing verification, corrective action, and risk-adjusted monitoring.
This guide presents a practical framework for transforming your supplier management approach from static qualification to dynamic assurance—connecting PQQ baselines, method-specific lab verification, structured audits, and disciplined CAPA into a unified Plan-Do-Check-Act cycle that both buyers and suppliers can execute from.
Why Pre-Qualification Alone Isn’t Enough
A typical pre-qualification questionnaire asks suppliers to confirm registrations, provide test reports, and describe their quality systems. The supplier returns a package of certificates and data sheets. The buyer reviews them, finds everything nominally in order, and approves the supplier.
Three months later, the first shipment arrives 15% over target moisture content, causing web breaks during conversion. The lab report the supplier provided during PQQ showed moisture at 7.2%—well within spec. What changed?
Often, nothing changed. The PQQ captured a moment in time under specific conditions. It didn’t establish how the supplier measures moisture (which ISO method, what sampling protocol, calibration frequency), who is responsible for monitoring drift, or what triggers corrective action when results fall outside control limits.
Pre-qualification establishes capability in theory. Ongoing assurance confirms capability in practice. The distinction matters because kraft paper manufacturing involves dozens of variables—fiber source, refining conditions, machine speed, ambient humidity—that can shift performance even when the production system itself remains stable.
The PDCA Loop: From Checkpoint to Continuum
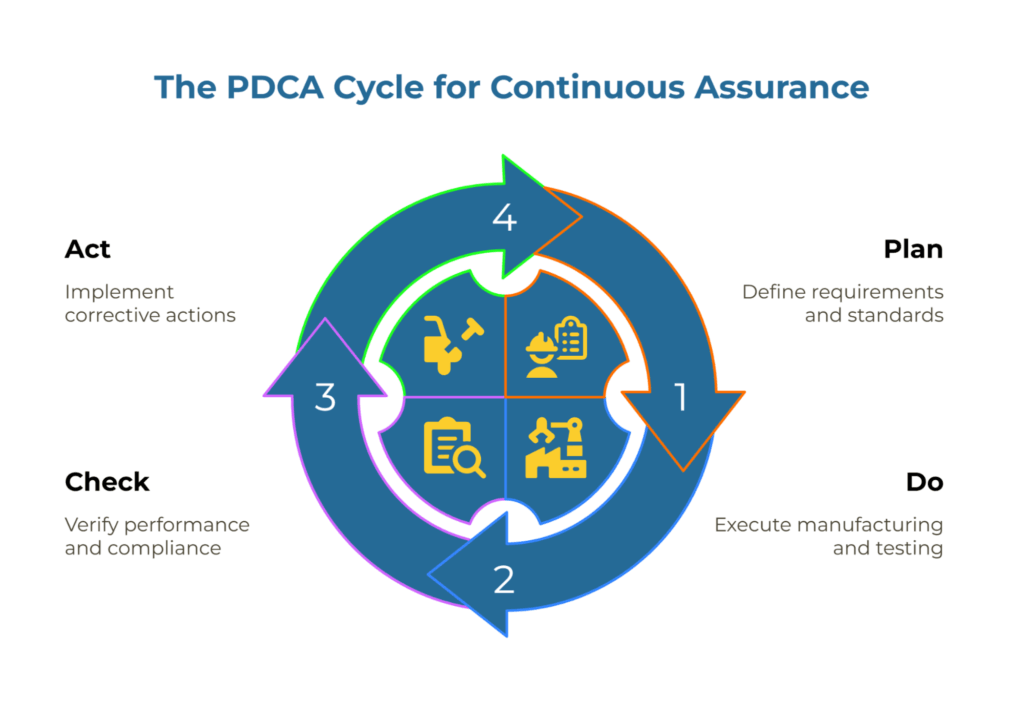
The Plan-Do-Check-Act framework provides the structure for continuous assurance. In this context:
Plan represents your PQQ requirements and the evidence standards you define upfront. You specify which test methods apply to each property, what documentation proves traceability, and which risk factors determine audit frequency.
Do covers the supplier’s execution—manufacturing to specification, testing using agreed methods, maintaining calibration records, and documenting deviations.
Check encompasses your verification activities: reviewing method-identified lab reports, conducting risk-appropriate audits (remote or on-site), and comparing claimed performance against actual delivery metrics like on-time-in-full rates.
Act closes the loop through structured CAPA when gaps appear, then feeding lessons learned back into updated PQQ requirements or revised monitoring cadences.
This isn’t bureaucracy for its own sake. It’s a decision framework that helps you allocate verification resources efficiently while giving suppliers clear performance standards and a pathway to demonstrate reliability over time.
Designing a PQQ That Feeds Audits (Not Just Paperwork)
An audit-ready PQQ focuses on evidence that can be verified later, not claims that sound reassuring but prove impossible to confirm. The difference shows up in how you structure questions and what documentation you require.
Minimum Evidence Set with Built-in Verifiability
Effective PQQs demand three categories of evidence:
Registration and certification scope. Request registration numbers for quality systems (ISO 9001), environmental management (ISO 14001), or chain-of-custody programs (FSC, PEFC). Critically, require the certificate’s scope statement and validity period. A mill might hold ISO 9001 certification, but if the scope excludes the specific product line you’re sourcing, the certificate provides no assurance for your purchase. During audits, you verify these registrations against public registries—tools like IAF CertSearch allow you to validate accredited certifications across multiple schemes and jurisdictions. Confirm the scope language matches your product requirements.
Product-specific test data with method identification. Generic claims like “high burst strength” mean nothing without numbers, units, and test standards. Your PQQ should require suppliers to report every property with its corresponding test method. For kraft paper, this typically includes moisture content (ISO 287), burst strength (ISO 2758), water absorption (ISO 535 Cobb test), and grammage (ISO 536). Auditors later verify that the supplier’s lab actually follows these methods by reviewing equipment lists, calibration certificates, and witnessing test procedures.
Traceability documentation. Request evidence of how the supplier tracks material from raw fiber through the finished product. This might include batch coding systems, production logs that link shift data to specific orders, or digital systems that tie individual test results to roll identifiers. During audits, you trace a sample backward through the production record to confirm the system works as described.
Buyer Verification vs. Supplier Demonstration: A RACI View
Confusion about who owns what often undermines the PQQ-to-audit connection. A simple RACI framework clarifies responsibilities:
| Activity | Buyer | Supplier | Auditor (if 3rd party) |
| Define required test methods | Responsible | Consulted | Informed |
| Perform product testing | Informed | Responsible | Accountable (verify) |
| Maintain calibration records | Informed | Responsible | Accountable (verify) |
| Report deviations | Informed | Responsible | Consulted |
| Verify scope/validity of certificates | Accountable | Informed | Responsible |
| Conduct on-site process audits | Accountable | Consulted | Responsible (if hired) |
| Define corrective action | Accountable | Responsible | Consulted |
The buyer sets requirements and verifies compliance. The supplier executes and provides evidence. When third-party auditors are involved, they perform detailed verification on the buyer’s behalf but don’t set the standards.
This division prevents two common failures: buyers who demand data they can’t interpret or verify, and suppliers who submit reports that look professional but lack the detail needed for meaningful assessment.
For more detailed guidance on structuring these requirements, see our PQQ template: minimum evidence required from kraft paper suppliers.
Method-Identified Lab Evidence: Making Reports Comparable
Two suppliers might both claim “8% moisture content,” but if one tested using ISO 287 at 23°C and 50% relative humidity while the other used an internal method at uncontrolled conditions, the numbers aren’t comparable. Method identification transforms subjective claims into verifiable data.
Pairing Properties with Standardized Test Methods
For kraft paper, several ISO and TAPPI standards provide internationally recognized testing protocols:
- ISO 287 specifies conditioning and moisture determination procedures, including the oven-dry method that serves as the reference for most paper testing.
- ISO 2758 defines burst strength testing using a diaphragm apparatus, critical for kraft grades used in demanding packaging applications.
- ISO 535 describes the Cobb test for measuring water absorption, essential when the end-use involves exposure to humidity or liquid content.
- ISO 536 establishes grammage (basis weight) measurement, the foundational property for specification and pricing.
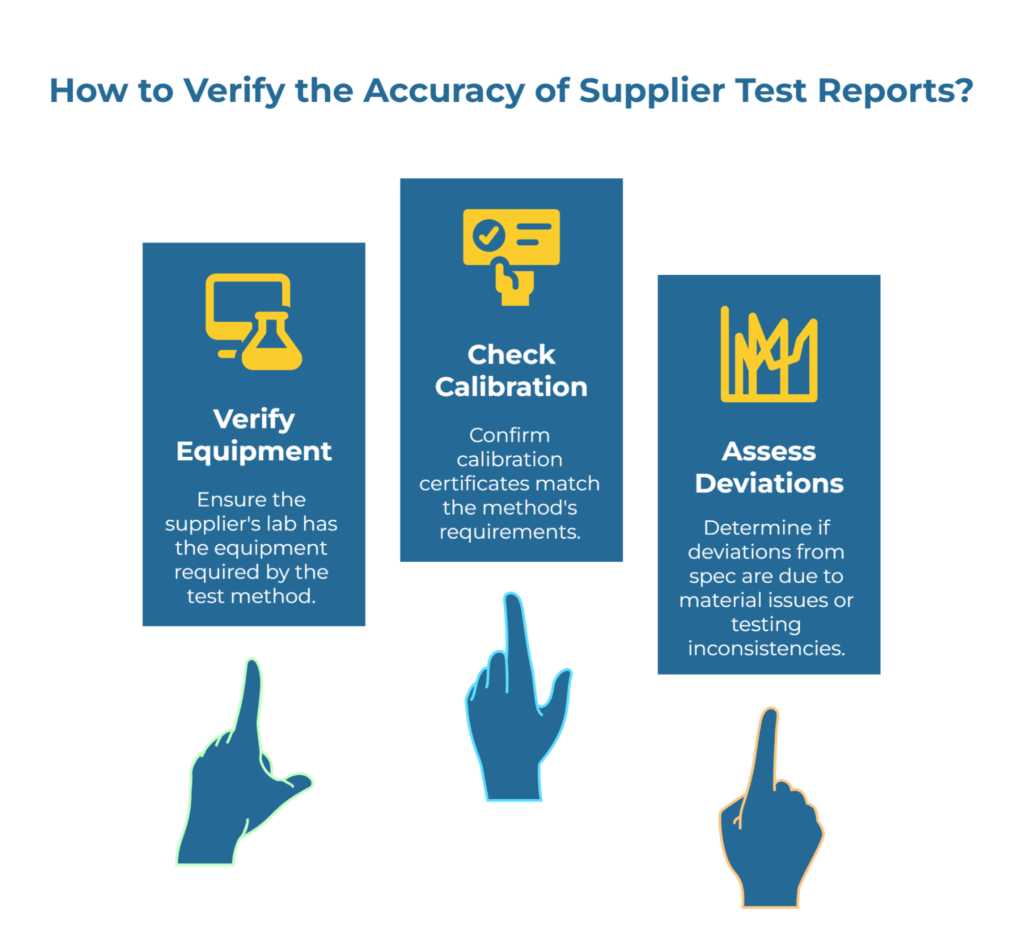
When you receive a test report, every value should include its method reference. Not “burst strength: 350 kPa” but “burst strength: 350 kPa per ISO 2758.” This seemingly small detail enables several critical verification steps during audits.
First, you can confirm the supplier’s lab actually has the equipment required by that method. ISO 2758 requires a specific diaphragm burst tester; if the lab is using a different apparatus, the results may not correlate.
Second, you can verify calibration certificates match the method’s requirements. ISO 287 moisture testing demands temperature control within ±2°C. If the supplier’s environmental chamber certificates show wider tolerances, their moisture data is questionable.
Third, you can assess whether deviations from spec represent true material issues or testing inconsistencies. When a supplier reports moisture just outside specification, knowing they use ISO 287 with proper calibration gives you confidence in the result. Unidentified methods leave you guessing.
The Moisture-First Principle

Moisture content deserves special attention because it affects nearly every other paper property. Most mechanical tests—burst, tensile, tear—show significant sensitivity to moisture levels. A sample tested at 9% moisture will show different burst strength than the same paper tested at 6% moisture.
This creates a verification sequence: confirm moisture is within specification and measured correctly before evaluating other properties. During audits, this often means witnessing moisture testing first, then checking whether conditioning procedures (bringing samples to standard atmospheric conditions) were properly followed before mechanical tests.
Suppliers sometimes report excellent burst or tensile results but reveal poor moisture control practices during audits. Those mechanical test numbers, however impressive, become unreliable without confidence in the underlying moisture measurement. For practical guidance on interpreting these reports, refer to how to interpret lab test reports for kraft paper.
Choosing Remote vs. On-Site Audits by Risk
Not every supplier requires the same verification intensity. A multinational mill with ISO 9001 certification, a ten-year track record, and consistently low defect rates needs different oversight than a new converter with limited references. Risk-tiering your audit approach allocates resources efficiently.
When Remote Audits Provide Sufficient Assurance

Remote audits—conducted via document review, video facility tours, and virtual interviews—work well when several conditions align:
The supplier has an established quality system certified to recognized standards and can provide evidence of ongoing compliance (surveillance audit reports, internal audit results).
Your order history shows stable performance with low defect rates, on-time delivery, and minimal customer complaints attributable to quality issues.
The product specification falls within the supplier’s core competency and published capabilities, involving no novel processes or materials.
Third-party test data or customer references corroborate the supplier’s quality claims.
Under these conditions, a remote audit can verify documentation, confirm key personnel understand requirements, and spot any obvious gaps in procedures without the cost and time of travel. Many organizations structure their remote audit approach following ISO/IEC TS 17012 guidance on using information and communication technology in conformity assessment. Many buyers use remote audits as their primary verification tool for established, low-risk suppliers, reserving on-site visits for periodic re-validation or when specific concerns emerge.
Triggers That Demand On-Site Verification
Certain situations require physical presence:
New supplier relationships. When you lack performance history, an on-site audit confirms the operation matches the documentation. You verify production equipment exists and functions, lab instruments are calibrated, and personnel demonstrate competence in procedures they’ve described on paper.
Prior non-conformances. If a supplier has delivered off-spec material or missed agreed specifications in previous orders, an on-site audit investigates root causes and verifies corrective actions. Document review alone cannot confirm whether a new calibration procedure is being followed consistently or whether retraining actually changed operator behavior.
Scope changes. When a supplier proposes to manufacture a new grade outside their historical product range, or when you’re considering them for a critical application that demands tighter tolerances, on-site verification confirms process capability before committing to large orders.
Regulatory or customer requirements. Some industries mandate on-site audits for supplier approval, particularly when the end product has safety implications or must meet stringent regulatory standards.
For more detailed decision criteria, consult remote vs. on-site supplier audits for kraft paper.
What a Complete Audit Report Must Answer

Regardless of format, every audit report should address four fundamental questions. Many organizations align their audit reporting structure with ISO 19011 guidelines for auditing management systems:
- Who conducted the audit and what was their scope? Identify the auditor (internal team, third-party firm), their qualifications, the departments and processes they reviewed, and any limitations on access.
- How did they verify claims? Document the audit methodology—whether they witnessed production runs, reviewed calibration certificates, interviewed personnel, traced materials through the system, or analyzed data records.
- What did they find? Present objective findings, distinguishing between major non-conformances (issues that could lead to defective product), minor non-conformances (documentation gaps or procedural deviations that don’t immediately affect quality), and observations (opportunities for improvement).
- What happens next? Specify required corrective actions, responsibility assignments, completion deadlines, and the follow-up mechanism to verify effectiveness.
Audit reports that merely state “facility passed inspection” or list generic observations provide little value. The detail level should enable you to make informed risk decisions and track corrective action through closure. Our checklist: what to require in a kraft paper supplier audit report offers a practical template for evaluating report completeness.
CAPA That Actually Closes the Loop
Finding problems is straightforward. Fixing them permanently is not. Many organizations implement corrective actions that address symptoms while leaving root causes intact, leading to recurrence. A structured CAPA process prevents this pattern.
The Four-Part CAPA Sequence
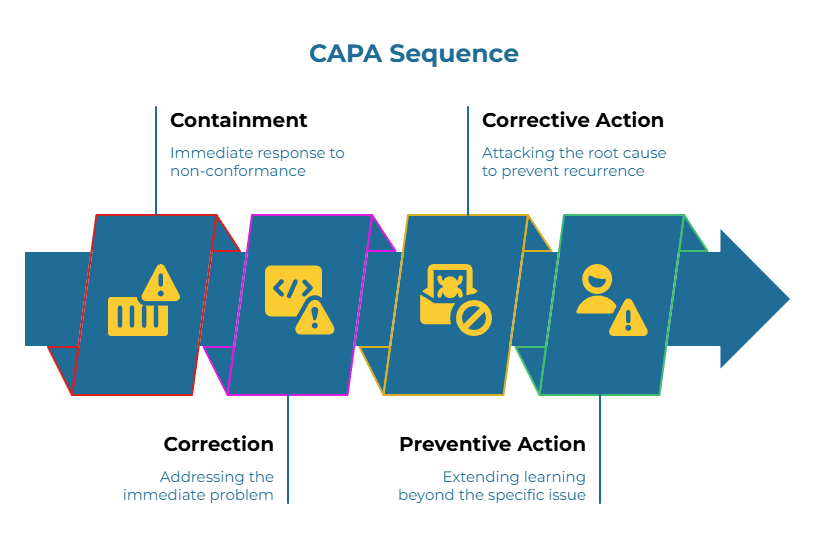
Effective corrective and preventive action follows a consistent pattern: contain, correct, verify, prevent.
- Containment represents your immediate response when a non-conformance appears. If a shipment arrives over moisture specification, containment might involve segregating affected material, implementing 100% inspection of remaining inventory from that lot, and holding further shipments pending investigation. The goal is preventing defective material from reaching your production line or end customers while you investigate.
- Correction addresses the immediate problem. In the moisture example, this might involve the supplier re-drying affected rolls and re-testing them using the agreed ISO 287 procedure with witnessed verification.
- Corrective action attacks the root cause to prevent recurrence. If investigation reveals the moisture deviation resulted from a malfunctioning humidity sensor in the supplier’s conditioning room, corrective action includes repairing or replacing the sensor, verifying proper function through calibration, and testing previously manufactured material to determine if the problem was isolated or systemic.
- Preventive action extends learning beyond the specific issue. Perhaps the investigation revealed that the supplier’s calibration schedule for environmental controls was inadequate. Preventive action updates the schedule, adds redundant sensors, and implements more frequent verification checks. It might also trigger a review of other environmental controls throughout the facility to confirm they receive adequate maintenance attention.
This four-part structure aligns with corrective action requirements found in quality management standards like ISO 9001 and in regulated industries—for example, the FDA mandates CAPA procedures under 21 CFR 820.100 for medical device manufacturers. The mechanics—documented cause analysis, effectiveness verification, and thorough record-keeping—apply broadly across manufacturing sectors.
Evidence Trails and Method Consistency
CAPA effectiveness depends on verifiable evidence, not promises. When a supplier reports corrective action complete, your verification should include:
- Evidence the immediate problem was resolved. Request test data showing the corrected material now meets specification, ideally using the same method (ISO 287 for moisture) to ensure comparability.
- Evidence the root cause was addressed. Obtain calibration certificates for repaired equipment, training records for retrained personnel, or process documentation showing updated procedures.
- Evidence the correction is holding. Review ongoing performance data over a reasonable period (often several production runs or a defined time window) to confirm the issue hasn’t recurred.
Method consistency matters here just as it did in initial qualification. If a supplier initially reported moisture using ISO 287, their post-correction verification should also use ISO 287. Switching methods during CAPA makes it impossible to determine whether improvement reflects genuine change or simply different measurement approaches. For detailed CAPA design principles specific to kraft paper operations, see designing CAPA workflows for kraft paper suppliers.
Operational Cadence and Escalation Triggers
A functioning assurance system requires clear rules about sampling frequency, acceptable quality levels, and the conditions that trigger more intensive oversight. These parameters prevent both under-reaction (missing systemic problems) and over-reaction (excessive inspection costs).
Risk-Adjusted Sampling
While detailed acceptance sampling theory involves statistical concepts beyond this guide’s scope, the general principle is straightforward: inspect more intensively when consequences of defects are severe or when supplier reliability is unproven, less intensively when the supplier has demonstrated consistent quality.
Many organizations use acceptance sampling by attributes with an Acceptable Quality Level (AQL)—the target defect rate used to design sampling plans. ISO 2859-1 provides internationally recognized procedures for establishing these sampling schemes based on lot size and acceptable risk levels.
For critical properties that could cause production failures or safety issues—burst strength in packaging for heavy goods, moisture content affecting dimensional stability—you might inspect every lot initially, reducing frequency only after the supplier demonstrates stable process control over multiple deliveries.
For less critical properties or established suppliers with excellent track records, periodic sampling may suffice, perhaps testing every third or fifth lot rather than every delivery.
The key is documenting your sampling plan explicitly and adjusting it based on performance. A supplier who consistently meets specification with minimal variation can earn reduced inspection intensity, while defects or near-misses trigger temporary increases until confidence is restored.
Escalation Triggers That Prevent Small Problems from Becoming Large Ones
Define specific conditions that require heightened attention:
- Repeat defects. If the same non-conformance appears twice within a defined period (often 90 days), escalate even if individual instances were contained. Recurrence suggests inadequate corrective action or ineffective verification.
- Calibration drift. When audit findings or received test data show the supplier’s measurement results diverging from third-party lab results or from their own historical patterns, investigate calibration systems before the drift produces out-of-specification material.
- Documentation discrepancies. Minor paperwork errors might not warrant escalation individually, but patterns of inconsistency—missing signatures, conflicting dates, test results that don’t match shipping documentation—signal control breakdowns that eventually produce quality issues.
- Threshold violations. Pre-define acceptable ranges for key metrics (on-time delivery, defect rates per thousand units, customer complaint frequency) and escalate automatically when thresholds are breached.
Escalation might involve moving from remote to on-site audits, increasing inspection frequency, requiring third-party verification, or placing the supplier on probationary status pending demonstrated improvement.
Putting It Together: A One-Page Assurance Workflow
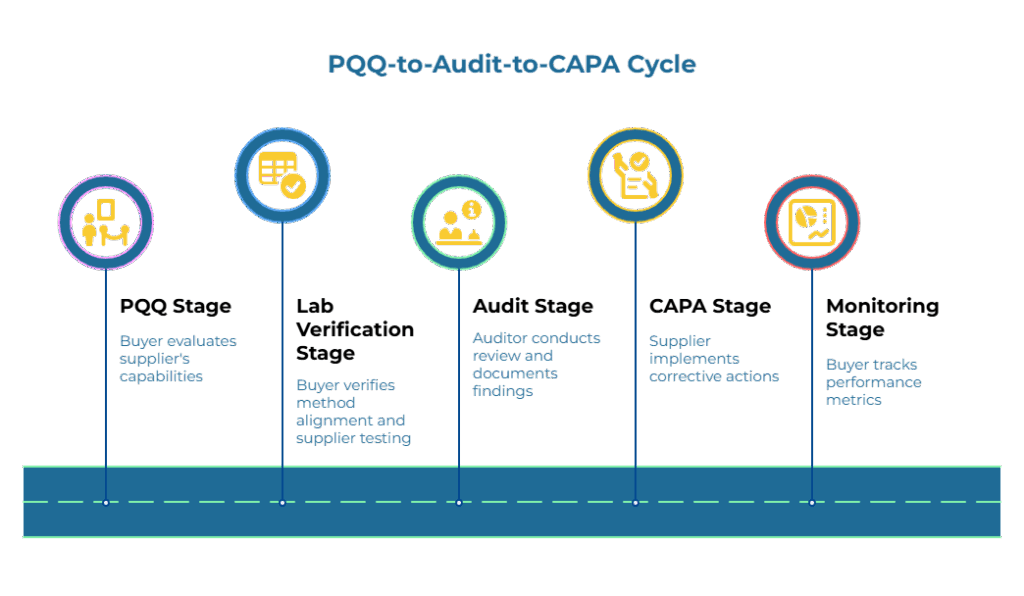
The complete PQQ-to-audit-to-CAPA cycle can be visualized as a continuous loop with defined inputs, responsible parties, and decision gates at each stage:
PQQ Stage:
- Input: Completed questionnaire with required evidence (certificates, method-identified test data, traceability documentation)
- Owner: Buyer evaluates; supplier provides evidence
- Artifact: Approved vendor record with documented baseline requirements
- Decision Gate: Does evidence demonstrate capability? If yes, approve for trial orders; if no, request additional information or decline
Lab Verification Stage:
- Input: Ongoing test reports from supplier, third-party validation results, received material inspection data
- Owner: Buyer verifies method alignment; supplier performs testing
- Artifact: Quality dashboard tracking trends in key properties (moisture, burst, Cobb, grammage)
- Decision Gate: Are results consistently within specification? If yes, maintain current sampling frequency; if trends show drift, trigger investigation
Audit Stage:
- Input: Performance data, risk assessment, audit schedule
- Owner: Auditor (internal or third-party) conducts review; supplier facilitates
- Artifact: Audit report documenting findings, non-conformances, and required actions
- Decision Gate: Are controls effective? If major non-conformances exist, implement CAPA and increase oversight; if minor issues, schedule follow-up verification; if no issues, maintain current approach
CAPA Stage:
- Input: Identified non-conformances or defects
- Owner: Supplier implements correction; buyer verifies effectiveness
- Artifact: CAPA documentation showing containment, correction, root cause, preventive measures, and effectiveness verification
- Decision Gate: Is corrective action effective? If yes, resume normal operations and update PQQ baselines if needed; if no, escalate and consider supplier status change
- Monitoring Stage:
- Input: Delivery performance metrics (OTIF rates, defect frequencies, customer feedback)
- Owner: Buyer tracks trends; supplier reports
- Artifact: Supplier scorecard with performance trends over time
- Decision Gate: Is performance stable or improving? If yes, maintain current cadence; if declining, investigate causes and adjust audit frequency
This workflow is not meant to be rigid bureaucracy. It’s a decision-support framework that helps you allocate verification resources based on risk and performance, ensuring attention goes where it’s needed most.
How PaperIndex Helps (Neutrally)
PaperIndex operates as a neutral connector between buyers and suppliers in the global paper and pulp industry. The platform provides two primary services relevant to implementing the assurance framework described in this guide.
First, the PaperIndex Academy offers practical resources covering supplier qualification, audit preparation, test method interpretation, and quality system design. These educational materials are freely accessible and designed to serve both buyers building procurement capabilities and suppliers working to meet customer requirements.
Second, PaperIndex’s supplier discovery tools allow buyers to find kraft paper suppliers based on capability criteria, geographic location, and product specifications. The platform does not engage in brokerage, price publishing, or market intelligence services—it functions solely to facilitate connections and provide educational support.
For organizations implementing evidence-based supplier assurance programs, these resources complement your internal processes by providing standardized templates, industry context, and access to suppliers who understand the quality expectations described throughout this guide.
Frequently Asked Questions
What should a kraft paper supplier audit report include?
A complete audit report should document the audit scope and timeframe, identify which process areas and production lines were reviewed, describe the verification methods used and samples examined, present findings categorized by severity (major non-conformances, minor non-conformances, observations), and attach supporting evidence such as records, images, and method-identified test results. Many organizations structure their audits following ISO 19011 guidelines for management system auditing to ensure consistent, thorough reporting.
When is a remote audit acceptable for kraft paper mills?
Remote audits work effectively when risk is low, the supplier has strong documented controls, and verification can be performed reliably through digital means—document review, video facility tours, instrument logs, and virtual personnel interviews. Organizations typically reserve on-site audits for higher-risk situations: new supplier relationships, repeat quality issues, significant process or grade changes, or when physical verification of controls is essential. The choice often follows risk assessment frameworks aligned with ISO/IEC TS 17012 guidance on using information technology in conformity assessment.
Which lab standards apply to kraft paper moisture and strength?
The primary ISO standards for kraft paper testing include ISO 287 for moisture content determination, ISO 2758 for bursting strength measurement, ISO 535 for water absorptiveness (Cobb test), and ISO 536 for grammage. TAPPI publishes parallel methods widely used in North America. The critical requirement is consistency—once you and your supplier agree on specific methods, all testing and verification should use those same standards to ensure results remain comparable over time.
How do I verify the scope and validity of certificates?
Certificate verification requires three steps. First, examine the certificate itself for the issuing standard, specific scope statement (which grades and processes are covered), and validity dates. Second, confirm the certification body’s legitimacy and accreditation status. Third, where available, validate the certificate through accredited directories such as IAF CertSearch, which provides searchable records across multiple certification schemes and jurisdictions. The scope verification is particularly critical—a kraft paper mill may hold ISO 9001 certification, but if the certificate scope excludes your specific product line, it provides no assurance for your purchase.
Glossary of Key Terms:
- AQL (Acceptable Quality Limit): The target defect rate used to design acceptance sampling plans, often following ISO 2859-1 procedures.
- CAPA (Corrective and Preventive Action): A structured process for addressing problems and preventing their recurrence, required by quality standards like ISO 9001 and regulatory frameworks like FDA 21 CFR 820.100.
- ISO (International Organization for Standardization): Body that publishes internationally recognized technical standards, including those for paper testing and quality management.
- OTIF (On-Time In-Full): A delivery performance metric measuring whether shipments arrive when promised and contain the correct quantities.
- PDCA (Plan-Do-Check-Act): A continuous improvement cycle used to establish and maintain quality systems.
- TAPPI (Technical Association of the Pulp and Paper Industry): Organization that publishes technical standards and testing methods for pulp, paper, and packaging.
Disclaimer: This is educational content. All examples are illustrative.
Our Editorial Process
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

