📌 Key Takeaways
Resilient corrugated box sourcing requires converting technical specifications into verifiable supplier evidence before awarding contracts—not just comparing unit prices.
- Evidence Gates Prevent Production Failures: Requiring quality system documentation, test capability proof, and traceability records at the RFQ stage surfaces capability gaps before they become line stoppages.
- Stage-Gated Pre-Qualification Filters Risk Early: A three-stage funnel—documentation review, sample/trial validation, and reference checks—eliminates suppliers who cannot consistently meet specifications at scale.
- Standardised RFQs Force True Comparability: Requiring identical test methods, delivery terms using Incoterms® 2020, and evidence annexes with quotes prevents apples-to-oranges bid comparisons.
- Risk-Adjusted Scoring Beats Price-Only Decisions: Weighting quality evidence strength, delivery reliability, and change control discipline alongside landed cost predicts supplier performance better than unit price alone.
- Post-Award Scorecards Maintain Compliance: Quarterly performance tracking with defined thresholds for delivery failures, quality rejections, and change control violations activates backup sourcing before crises occur.
Spec-to-sourcing systems convert compliance requirements into defensible supplier decisions.
Procurement managers, packaging engineers, and quality leads in regulated industries will gain a repeatable verification framework here, preparing them for the stage-by-stage implementation roadmap that follows.
The pallet arrived again. The cartons feel slightly damp, and the report you expected is missing. You can already see the chain reaction: receiving QC flags the lot, production asks for a workaround, and Procurement gets pulled into a blame loop that has nothing to do with negotiating price.
Most buyers treat corrugated box sourcing as a basic price negotiation. But in regulated industries like pharma, it’s a risk-control process. A resilient sourcing strategy depends on three things: a standardized mill spec sheet that defines strength, material grades, and testing tolerances; supplier verification that confirms a manufacturer can consistently meet those standards at scale; and compliance-aligned procurement workflows that prevent mismatches between purchase orders, labels, and audit documentation. When any of these links break, the result isn’t just cost creep—it’s quarantine holds, compliance findings, and real operational downtime.
The spec is done. Engineering signed off.
Now what?
The conference room empties. The PDF gets emailed to procurement with a note: “Ready for sourcing.” But that handoff—spec complete to supplier selected—is where resilience either gets built or quietly breaks down. A spec is a contract with engineering; sourcing is a contract with reality. And reality has a habit of exposing gaps that look fine on paper.
This article provides a roadmap for that translation layer: converting technical specifications into verifiable sourcing criteria, building a stage-gated supplier qualification funnel, and designing evaluation systems that account for risk—not just price. By the end, procurement professionals, packaging engineers, and quality leads should have a repeatable framework for making supplier decisions that hold up under production pressure and audit scrutiny.
Workflow at a Glance
Requirements intake → measurable spec (test method + acceptance criteria) → sourcing criteria + evidence checklist → shortlist → pre-qualify (documentation gate → samples/trials gate → references gate) → standardized RFQ + evidence annex → risk-adjusted scoring → pilot/first run validation → contract + SLA + change control → ongoing scorecard monitoring.
Operational Vulnerabilities Post-Specification
Specifications tell you what you need. They don’t tell you how to verify that a supplier can deliver it consistently.
Three failure modes dominate this translation layer:
Ambiguous acceptance criteria create supplier disputes. When sourcing corrugated boxes, the spec says “burst strength ≥1200 kPa” but doesn’t name the test method (ISO 2758 vs. TAPPI T 403), conditioning protocol, or sampling plan. Two suppliers quote the same number using different methods. One passes incoming QC. One doesn’t. The dispute consumes weeks.
Missing evidence expectations delay leverage. Procurement requests quotes. Suppliers send prices. Nobody asks for quality system documentation, test capability proof, or traceability records until after the first shipment fails. By then, the leverage is gone—and the line stoppage has already happened.
Siloed decision ownership creates handoff gaps. Engineering writes the spec. Procurement owns the award. QA owns incoming inspections. Nobody owns the handoff. The category manager fears selecting a supplier on price only to face quality failures or compliance risk. The operations manager fears boxes arriving late or failing strength tests, disrupting packaging lines. Both fears trace back to the same root: a process that doesn’t require evidence at each stage.
If the vendor cannot document their quality system, testing standards, and compliance readiness, then even a perfect quote becomes a risk multiplier.
This isn’t about finding the “best supplier.” That framing is part of the problem. It’s about building a system that surfaces capability gaps before they become production problems.
The Spec-to-Sourcing Scorecard
A supplier quote is a promise. The evidence pack is the proof.
The Spec-to-Sourcing Scorecard bridges technical requirements and supplier selection by defining what “qualified” actually means across multiple dimensions. Each dimension functions as a resilience lever—weakness in any one creates exposure that unit price alone cannot offset.
| Dimension | What to Request | What Adequate Evidence Looks Like |
| Performance & Fit | Test reports matching your spec’s named method and tolerance | Results within spec from an accredited or calibrated lab, with clear test conditions documented |
| Quality System Maturity | ISO 9001 and/or ISO 15378 (Primary packaging materials for medicinal products) certificates, internal QC procedures, calibration records, nonconformance/CAPA approach | Current certification with scope covering your product category, documented procedures, traceable calibration, clear corrective action discipline |
| Testing Capability | Lab capability list with example reports using defined methods | Capability matches critical tests; reports are traceable and method-aligned |
| Capacity & Continuity | Production capacity data, lead time commitments, contingency documentation | Capacity headroom sufficient for demand variability, documented backup plans for disruptions, realistic lead times with defined escalation |
| Compliance & Traceability | Lot traceability system, change control process, relevant regulatory certificates | Full batch-to-raw-material traceability, formal change notification procedures, COA/COC examples where applicable |
| Change Control Discipline | Change notification workflow and approval requirements | No silent substitutions; defined triggers, notice periods, and documentation requirements |
| Commercial Terms & Total Cost | Total landed cost breakdown, payment terms, service-level commitments | Transparent cost structure with same Incoterm basis, clear SLAs with defined remedies |
The ISO 20400 standard on sustainable procurement emphasizes that procurement decisions should consider the full lifecycle of supplier relationships—not just transaction price. This scorecard operationalizes that principle for corrugated packaging.
For detailed guidance on building the technical foundation, see creating your mill spec sheet: a guide for converting operations managers.
Translate Specifications into Sourcing Criteria
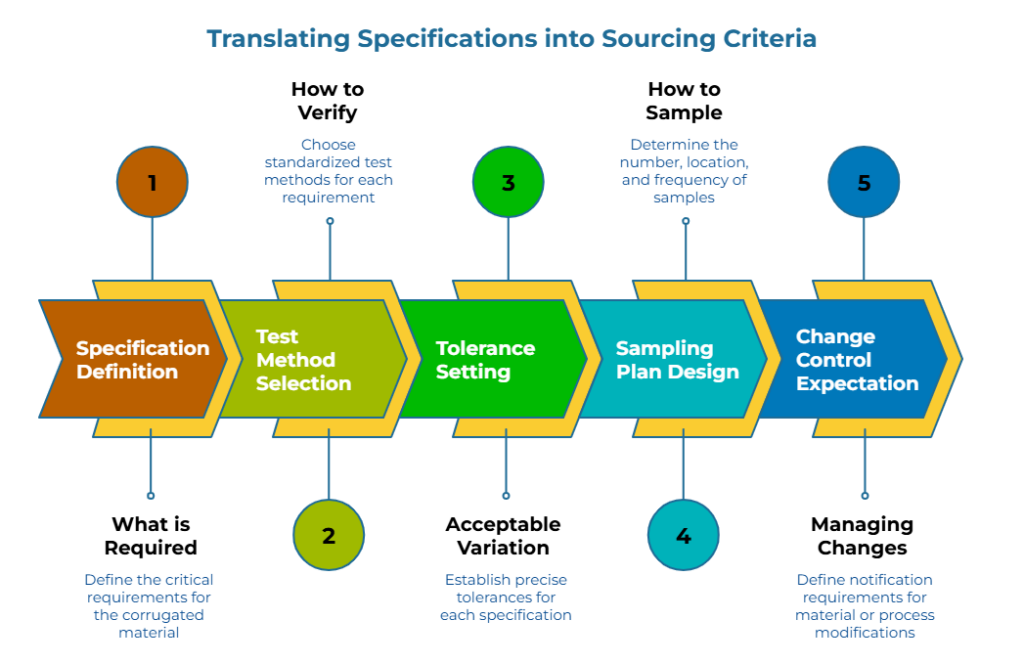
A spec says what. Sourcing criteria say how verification happens.
Most corrugated sourcing failures come from spec ambiguity and supplier inconsistencies—not the price itself. The translation process converts each critical requirement into four components:
The test method. Not “burst strength” but “burst strength per ISO 2758, conditioned per ISO 187.” Method differences can produce 5-15% variation in reported values for identical materials—enough to turn a compliant supplier into a rejection.
The tolerance. Not “approximately 200 gsm” but “200 gsm ±3%.” Vague tolerances invite interpretation. Interpretations invite disputes.
The sampling plan. How many samples, from which production lots, at what frequency. A supplier who passes on cherry-picked samples may fail on random production pulls.
The change control expectation. What requires notification before implementation—material substitution, process changes, equipment updates. Undisclosed material or process modifications frequently trigger performance erosion, compromising long-term consistency. Board grade drift without notice is a known corrugated failure mode: the quote looked fine, but receiving QC rejected the delivery.
If it can’t be verified, it can’t be managed.
This verification-first thinking aligns with risk management principles outlined in ISO 31000, which emphasizes that risk treatment should be based on explicit criteria rather than assumptions.
Build a Supplier Pre-Qualification Funnel
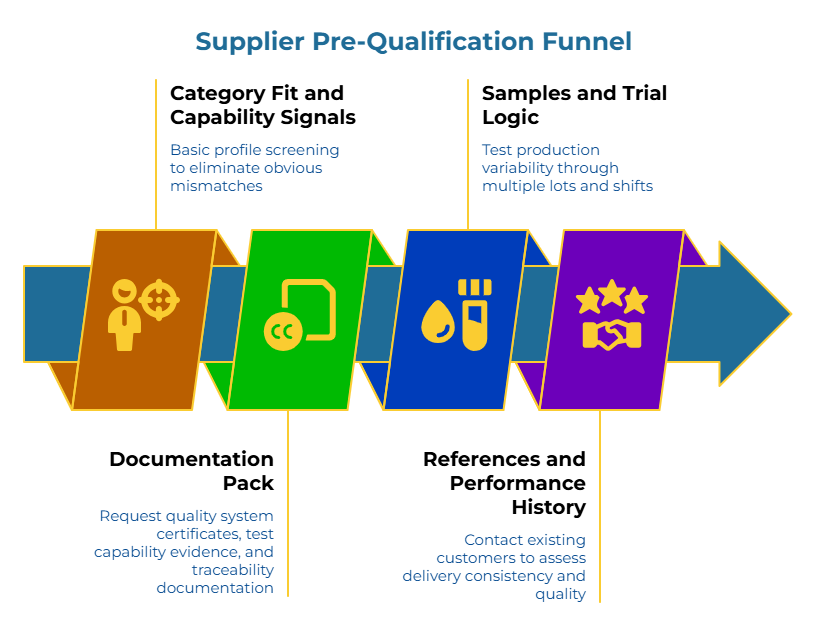
Pre-qualification filters out suppliers who can’t meet requirements before they consume RFQ bandwidth. The funnel structure prevents a common misconception: that certificates alone prove capability.
Stage 0: Category fit and capability signals. Does the supplier manufacture corrugated boxes in the grades, sizes, and volumes needed? Do they serve the relevant industry vertical (pharma, food, industrial)? Basic profile screening eliminates obvious mismatches early. For suppliers seeking to understand buyer requirements and connect with active procurement teams, exploring corrugated box buyers posting RFQs can provide valuable market intelligence on specification trends and sourcing patterns.
Stage 1: Documentation pack (evidence-first gate). Request quality system certificates, test capability evidence, and traceability documentation. Certificates prove a system exists. Test reports prove it functions. No documentation, no advancement. This stage addresses the operations manager’s core desire: suppliers who deliver consistently and meet performance standards.
Stage 2: Samples and trial logic. Samples prove capability on a single run. They don’t prove consistency. Design trials that test production variability—multiple lots, different shifts, real production conditions.
Watch for the sample-to-run gap. This is the difference between what a supplier achieves when trying to impress and what they deliver at scale. Sample approval does not guarantee production consistency—a misconception that causes repeated failures. A common surprise is moisture variability: two suppliers can meet the “same spec,” but boxes behave differently on the line because moisture control and storage practices vary.
Stage 3: References and performance history. Contact existing customers. Ask about delivery consistency, quality drift over time, responsiveness to problems. Past performance predicts future performance more reliably than promises or proposals.
To identify potential suppliers for the shortlist, PaperIndex provides access to a directory of corrugated box suppliers operating across major industrial markets. The platform functions as a discovery and contact layer—buyers send inquiries directly to suppliers, with all negotiations handled between the parties.
Design an RFQ That Forces Comparability
Most RFQs fail because they allow suppliers to quote on different assumptions. This creates non-congruent data sets that obstruct objective evaluation that delay decisions and create stakeholder conflict between procurement, engineering, and finance.
A standardized bid sheet fixes this by requiring identical fields, same quantity breaks, same delivery terms with specified Incoterms® 2020 basis, and same packaging assumptions. Suppliers must quote against the spec’s named test methods, not their internal standards.
The evidence annex requirement changes the dynamic entirely. The quote submission must include: quality system certificate (current), test capability evidence, sample test reports using the specified methods, and traceability documentation. Require the proof with the offer—not after award when leverage disappears.
Clear award criteria, communicated upfront, prevent post-submission renegotiation theater. Suppliers who understand how decisions get made submit more accurate, comparable quotes.
For more on structuring effective RFQs, see why traditional RFQs fail: the hidden cost of opacity in pharma packaging procurement.
Evaluate Beyond Unit Price
Unit price is one input. It’s not the decision.
Risk-adjusted evaluation addresses the category manager’s core fear: selecting a supplier based on price only to face quality failures, compliance risk, or missed deliveries. A weighted scoring model distributes emphasis across factors that actually predict supplier performance:
- Total landed cost (illustrative weight: 30%) — Normalize all quotes to the same Incoterm and delivery point before comparison
- Quality evidence strength (illustrative weight: 25%) — Score based on documentation completeness, test method alignment, and lab accreditation
- Delivery reliability (illustrative weight: 20%) — Score based on references, historical on-time-in-full data where available
- Continuity risk (illustrative weight: 15%) — Score based on capacity headroom, backup plans, financial stability signals
- Service responsiveness (illustrative weight: 10%) — Score based on RFQ response quality, communication clarity during qualification
These weights are illustrative. Each organization should calibrate based on their specific risk tolerance and operational priorities.
Risk multipliers adjust effective cost. A supplier with weak change control increases real cost through quality drift, rework, and audit exposure—even if their unit price is lowest. Scenario testing strengthens evaluation: How does each supplier perform under a demand surge? A supply disruption? An unannounced audit?
Close the Loop After Award
Selection isn’t the finish line. Post-award systems determine whether resilience holds over time.
Onboarding checklist. Confirm all documentation is current and matches what was submitted during qualification. Align on communication protocols, order processes, and escalation paths. Verify that incoming QC procedures match the spec’s test methods exactly.
Receiving QC and escalation. Define what gets tested, how often, and what triggers rejection versus concession. Document the escalation path for quality failures—who gets notified, what remedies apply, when dual-sourcing activates. This directly addresses line stoppage prevention concerns.
Quarterly scorecard. Track delivery performance, quality metrics, responsiveness, and compliance. Share results with the supplier. Use data to drive improvement—or justify switching. The scorecard creates accountability that prevents the gradual drift that degrades supplier performance over time.
Change control enforcement. Require notification before material substitutions, process changes, or equipment updates. Silent changes are a leading cause of the quality drift that makes spec-compliant suppliers fail in production.
Triggers for dual-sourcing or switch. Define the thresholds—consecutive late deliveries, quality rejections above a percentage, failure to notify changes—that activate backup sourcing or supplier replacement. Documented triggers remove ambiguity from difficult decisions.
For cross-functional alignment on these processes, see aligning procurement and engineering: a shared checklist for corrugated box RFQs.
Frequently Asked Questions
What evidence should be required before RFQ?
At minimum, require a documentation pack that demonstrates test capability, traceability discipline, and change control approach. Without that, the RFQ becomes a negotiation over assumptions.
Do certificates alone prove capability?
Certificates can be useful signals, but they rarely show whether the supplier can meet defined acceptance criteria at scale. Treat certificates as a starting point, then request process evidence and example test reports.
How should sample approval be handled?
Treat samples as validation, not a guarantee. Define the sample-to-run gap: critical-to-quality attributes, tolerances, and re-test triggers.
What is the simplest way to prevent apples-to-oranges quotes?
Use a standardized bid sheet and an evidence annex with the quote. If the evidence is missing, the bid is incomplete. One rule, big effect.
Adopt the Roadmap
Sourcing is not just buying a box. It’s selecting a compliance-ready partner who can meet measurable standards repeatedly.
The roadmap condenses to six stages:
- Convert specs into measurable criteria with named test methods, defined tolerances, and sampling plans
- Define evidence requirements before requesting quotes—documentation, test reports, traceability proof
- Pre-qualify suppliers through staged verification: documentation review, sample/trial validation, reference checks
- Issue standardized RFQs that force comparability and require evidence with the offer
- Evaluate with risk-adjusted scoring that weights quality evidence and continuity risk alongside price
- Close the loop with onboarding verification, SLAs, quarterly scorecards, and change control enforcement
Start with one category. Pilot the workflow. Measure the results: fewer quality failures, faster decisions, reduced rework loops between procurement, engineering, and QA. Cross-functional alignment shortens cycle time by eliminating the back-and-forth that plagues siloed processes.
Then scale.
Explore additional procurement frameworks, checklists, and templates at PaperIndex Academy.
Disclaimer:
This article provides general guidance on procurement practices and does not constitute legal, regulatory, or professional advice. Organizations should adapt these frameworks to their specific operational requirements, industry regulations, and risk tolerance. Examples and illustrative weights are provided for educational purposes and may require adjustment based on context.
Our Editorial Process:
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team:
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

