📌 Key Takeaways
Production lines stop when supplier issues surface mid-run—regular audits catch these problems before they become costly emergencies.
- Evidence-First Audits Prevent Line Stoppages: Systematic reviews of lab test conformance, certificate validity, and corrective action workflows surface small process drifts before they cross critical thresholds that cause press failures, web breaks, or adhesion problems on your production line.
- Test Method Alignment Eliminates False Rejections: Different testing standards like ISO 2758 versus TAPPI T 810 for burst strength produce incomparable results on identical material, creating unnecessary reject cycles when suppliers and buyers aren’t aligned on moisture determination (ISO 287), grammage measurement (ISO 536), and Cobb water absorption methods (ISO 535 or TAPPI T 441).
- Observable Red Flags Predict Future Quality Issues: Vague test reporting without method specifications, unnaturally perfect numbers showing no statistical variance, moisture readings consistently out of control at receipt, and slow paperwork arriving after shipments all signal suppliers who lack robust quality systems.
- Structured Qualification Beats One-Off Decisions: Pre-qualification audits validating basic capabilities, first-run audits within 30-60 days confirming real-world performance, and quarterly routine reviews for established suppliers transform supplier management from reactive crisis response to preventive risk control.
- Weighted Scorecards Drive Accountability: Tracking quality metrics (40%), documentation completeness (20%), service reliability (20%), and commercial stability (20%) with quarterly reviews identifies performance trends before they escalate into production stoppages or compliance failures.
Audits cost less than a single unplanned stoppage—they’re preventive insurance that catches certificate lapses, documentation errors, and process drift while they’re still manageable.
Procurement managers, quality assurance teams, and operations leaders at packaging manufacturers will find this framework essential, preparing them for the detailed audit protocols and field checklists that follow.
Production lines don’t stop on a schedule. They halt when a critical defect surfaces mid-run—often traced back to a supplier issue that could have been caught weeks earlier. For procurement and quality assurance managers at packaging manufacturers, this reactive cycle creates unnecessary risk and cost.
Regular, evidence-first supplier audits covering lab test conformance, certificate validity, and corrective action closure systematically surface small process drifts before they become line-stopping defects. This approach transforms supplier management from crisis response to preventive control, helping teams maintain stable production flows.
PaperIndex is a neutral, non-transactional connector serving the global pulp and paper industry. We don’t broker deals, sell market intelligence products, or publish pricing indices. Our role is to connect buyers with suppliers and provide educational resources that support informed decision-making.
What a “Real” Supplier Audit Covers (and Why It Prevents Stoppages)
The Definitional Frame—What Counts as a Kraft Paper Supplier Audit
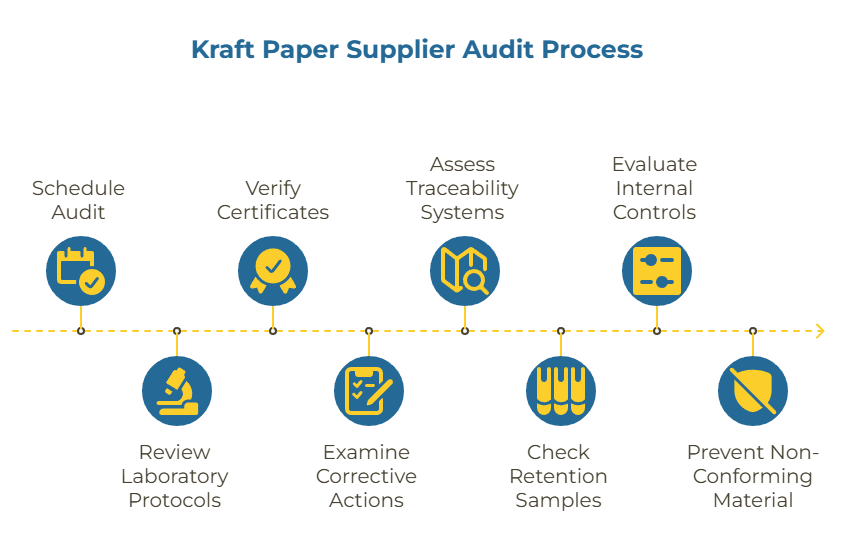
A kraft paper supplier audit is a scheduled, evidence-first review that examines three critical areas: laboratory testing protocols against recognized standards, active validity of quality and compliance certificates, and documented corrective action workflows. This differs from a simple facility tour or relationship check-in. The audit verifies that a supplier’s quality systems can consistently deliver material that meets your specifications under normal operating conditions.
The scope extends beyond production capability. It includes traceability systems, retention sample procedures, and the supplier’s internal controls for catching deviations before shipment. When these elements are verified through structured audits, they create multiple checkpoints that prevent non-conforming material from reaching your receiving dock.
How Small Drifts in Basis Weight, Moisture, Cobb, and Burst Become Downtime If Never Audited
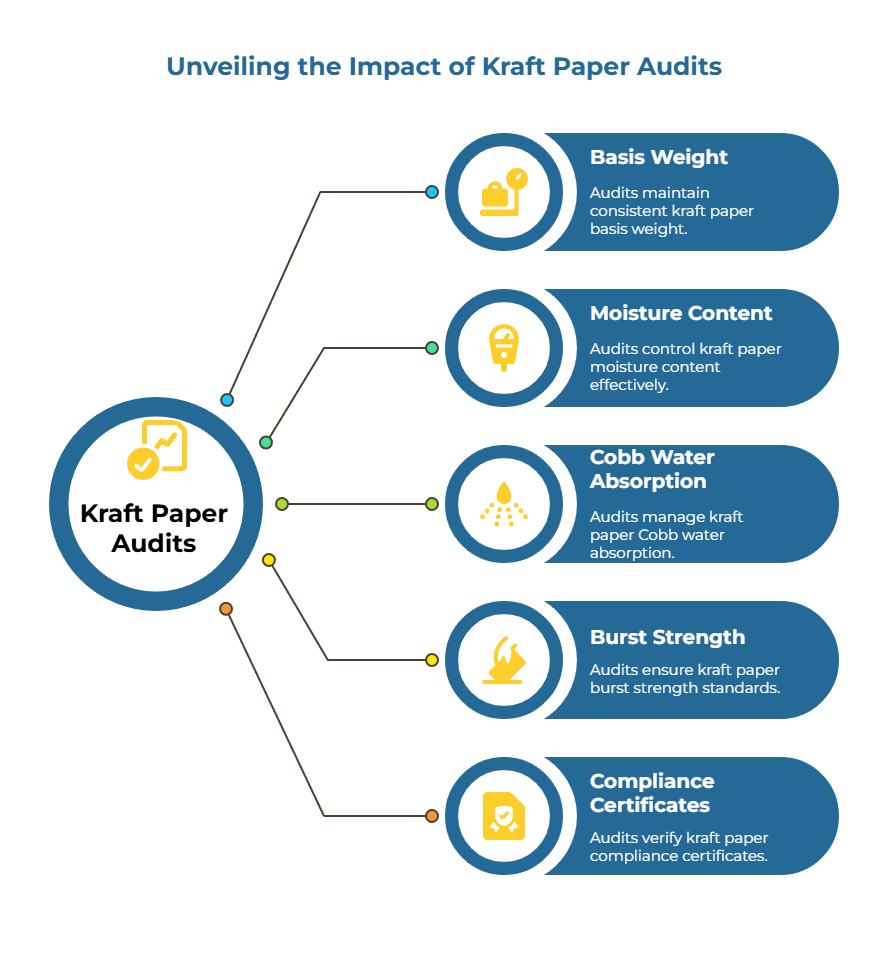
Kraft paper specifications define tight tolerances for basis weight, moisture content, Cobb water absorption, and burst strength. A supplier might gradually drift outside these parameters due to equipment wear, raw material variability, or process adjustments. Without regular audits, these small drifts accumulate undetected until they cross a critical threshold that causes press failures, web breaks, or adhesion problems on your production line.
For instance, if a supplier’s moisture control starts trending upward by 0.5% per month, it may still fall within the stated specification range for several months. However, by the time it triggers a quality complaint, you’re dealing with an immediate production stoppage rather than a manageable process adjustment. Verify FSC/ISO/food-contact certificates without missing scope to ensure all compliance requirements remain current and aligned with your operational needs.
The Root Causes of Downtime That Audits Catch Early
Certificate Lapses and Process Non-Conformance
ISO 9001 certificates, FSC or PEFC chain-of-custody credentials, and food-contact approvals all carry expiration dates and defined scopes. A supplier’s ISO 9001 certificate might be current, but its scope may not explicitly cover the kraft paper grades you’re purchasing. Similarly, an FSC certificate could lapse without immediate notice to buyers, creating compliance gaps that halt shipments when customs or your own quality systems flag the issue.
Process non-conformance occurs when a supplier’s actual manufacturing steps deviate from the documented procedures that were validated during initial qualification. These deviations often emerge gradually—a substituted raw material source, a modified drying temperature, or a bypassed quality checkpoint. Regular audits compare documented procedures against observed practices, catching these drifts before they manifest as defects in your facility.
Lab-Report Inconsistencies and Test Method Mismatches
Different testing standards produce different numerical results for the same physical property. A supplier using ISO 2758 for burst strength measurement will report values that aren’t directly comparable to TAPPI T 810 results, even when testing identical material. This creates false accept/reject cycles where material is rejected for being “out of spec” when the real issue is test method misalignment.
The most common areas requiring alignment include moisture content determination using ISO 287, grammage measurement per ISO 536, water absorptiveness via ISO 535 or TAPPI T 441, and burst strength through TAPPI T 810. Audits verify that suppliers are using the agreed-upon test methods specified in your purchase orders and that paper samples are properly conditioned to the standard atmosphere before testing to avoid skewed readings caused by off-condition stock.
They also check calibration records for testing equipment and confirm that lab technicians are properly trained. When these elements are validated through structured reviews, the resulting data becomes reliable for process control decisions rather than a source of confusion and disputes.
Documentation and Export Errors That Delay Materials
Bills of lading, certificates of origin, and fumigation certificates must match exactly across all documents in an international shipment. Prevent documentation-driven delays with a field-by-field export checklist that covers the critical data fields customs authorities scrutinize. A single discrepancy—wrong Incoterm, missing seal number, or mismatched container identifier—can trigger a hold at the port, effectively creating production downtime even though the material itself meets all quality specifications.
Supplier audits review documentation workflows and verify that the personnel preparing export paperwork understand the requirements for your destination country. This operational downtime prevention is as important as quality verification, particularly for packaging manufacturers operating on just-in-time inventory models.
Observable Warning Signs That Predict Quality Issues
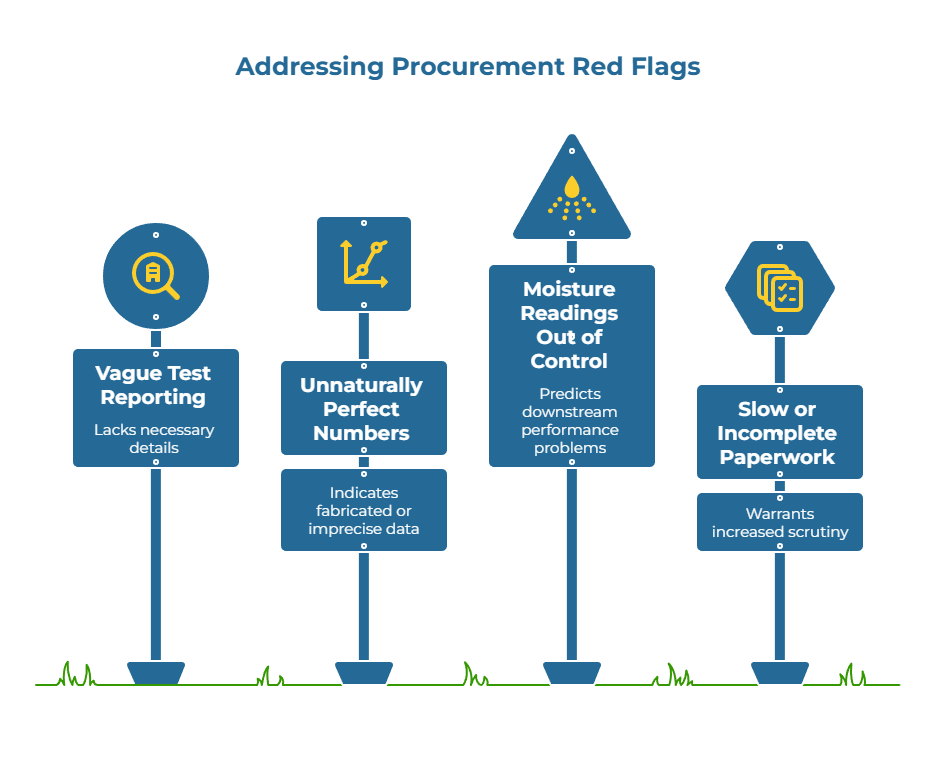
Experienced procurement teams learn to recognize patterns that signal future problems before they escalate. Vague test reporting such as “Cobb OK” without specifying the dwell time or standard used is a red flag—Cobb test results vary significantly depending on whether 60-second or 120-second contact time was used.
Another concerning pattern is unnaturally perfect numbers showing no variance across reels or months. Real manufacturing processes exhibit natural statistical spread; consistent identical values often indicate either fabricated data or inadequate measurement precision. Similarly, moisture readings that are consistently out of control at receipt relative to your storage climate predict downstream problems with Cobb performance, dimensional stability, and print quality.
Slow or incomplete paperwork that arrives after the shipment, or one-off capacity promises during peak seasons without documented redundancy plans, also warrant increased scrutiny during audits.
A Practical Audit Cadence for Kraft Paper Suppliers
Pre-Onboarding → First-Run → Quarterly/Biannual
The audit cycle begins before any purchase orders are issued. A pre-qualification audit validates that a potential supplier has the basic systems and capabilities required to meet your specifications consistently. During this phase, request three to five production-grade rolls or sheets from candidate lots, condition them properly, and test them via your own lab or a third party to validate performance claims. This initial assessment establishes a baseline for future evaluations and identifies any immediate red flags that should prevent onboarding.
After a supplier passes pre-qualification and you place trial orders, conduct a first-run audit within 30 to 60 days of receiving initial shipments. This audit verifies that the supplier’s performance under real production conditions matches what was observed during pre-qualification. It catches issues related to scaling up from sample production to commercial volumes. Approve the supplier only when both lab and line trials meet your thresholds for three consecutive lots, and paperwork is consistent.
For established suppliers with good track records, quarterly or biannual routine audits maintain oversight without creating excessive administrative burden. The frequency should increase if you observe declining on-time delivery rates, increasing quality complaints, or other performance indicators that suggest process drift. Choose the lower-risk partner: mills vs converters based on your specific risk tolerance and supply chain requirements.
Remote vs On-Site—When Each Is Appropriate
A remote desk audit reviews documents, test reports, and certificates without requiring travel to the supplier’s facility. This approach works well for routine checks of established suppliers who have demonstrated consistent performance. Request updated certificates, recent lab test summaries, and corrective action logs. Review these documents against your specifications and flag any discrepancies for follow-up.
Escalate to an on-site audit when remote signals indicate elevated risk. First-run failures, scope questions about certificates, process capability concerns, or significant changes to the supplier’s operations all warrant physical verification. On-site audits allow direct observation of manufacturing processes, inspection of testing equipment calibration, and confirmation that documented procedures reflect actual practices.
Field Checklist—What to Request, Verify, and Score
Evidence Package: Certificates, Lab Summaries vs Spec, CAPA Log, OTIF History
Begin each audit by requesting a complete evidence package before the audit date. This should include current ISO 9001 certificates with clearly defined scope statements, active FSC or PEFC certificates if required, any food-contact approvals relevant to your applications, and the most recent third-party audit reports if available.
Request at least three months of laboratory test summaries showing results for the specific kraft paper grades you purchase. These reports must specify the test methods used—such as ISO 287 for moisture, ISO 536 for grammage, ISO 535 or TAPPI T 441 for Cobb, and TAPPI T 810 for burst strength—along with the actual test conditions and lot-level values, not just pass/fail indicators. Compare these results directly against your purchase order specifications to identify trends or outliers.
The documentation package should also include detailed traceability data linking finished rolls to specific reel or pallet numbers, production machine and shift identifiers, and batch codes. Request change notifications for any modifications to fiber furnish, sizing chemistry, or calendering settings that could affect Cobb values or strength properties. The CAPA log documents how the supplier responded to previous quality issues, including root cause analysis and implemented fixes. On-time in-full delivery history reveals operational reliability beyond just quality metrics.
Shop-Floor Checkpoints: Capability Signals, Traceability, Retention Samples
During on-site audits, observe the actual manufacturing process to verify process capability. Look for statistical process control charts posted at critical control points, evidence of regular equipment calibration, and clear documentation of standard operating procedures. Ask to see traceability systems that link finished rolls back to specific raw material lots and production batches.
Retention sample procedures indicate quality consciousness. Suppliers should maintain representative samples from each production batch for a defined period, allowing retrospective analysis if quality issues emerge weeks or months after shipment. Set AQLs that minimize rejection risk post-audit by establishing clear acceptance quality limits based on defect criticality.
Pass/Fail Gates and Systematic Scoring
Establish clear scoring criteria before the audit. Define which findings constitute immediate fail conditions—such as expired critical certificates, absence of calibrated testing equipment, or inability to demonstrate traceability. These should trigger immediate corrective action requirements and potentially suspend shipments until resolved.
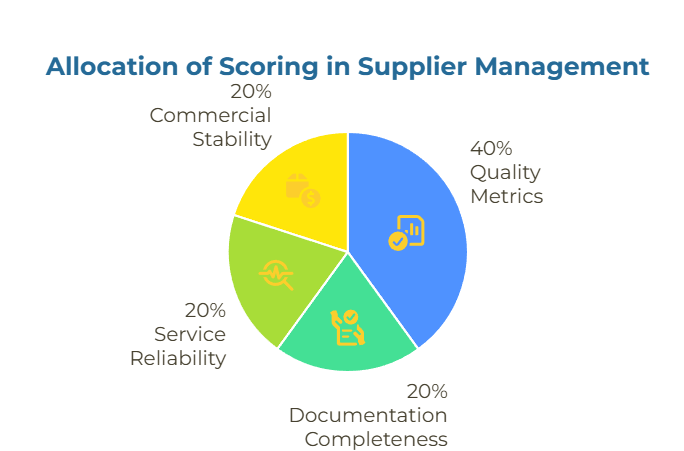
For ongoing supplier management, consider implementing a weighted scorecard approach that tracks performance across multiple dimensions. A practical framework allocates scoring as follows: Quality metrics including pass rates against tolerances and variance trends by lot (40%), Documentation completeness covering on-time delivery of traceable certificates of analysis (20%), Service reliability measured through lead-time adherence and communication responsiveness (20%), and Commercial stability encompassing price consistency and workable minimum order quantities (20%). Review these scores quarterly to identify trends before they become critical issues.
Lesser findings can be scored and accumulated. A supplier might pass the overall audit while still receiving action items to address. Define specific thresholds that trigger follow-up audits. For example, three medium-severity findings in a single area might require a focused re-audit of that process within 90 days. Use an evidence-first capability matrix for consistent supply to systematically evaluate suppliers across multiple dimensions.
Turning Findings Into Fewer Stoppages (Close the Loop)
CAPA Workflows and Re-Audit Triggers
Audit findings become preventive controls only when they drive actual changes in supplier behavior. Each finding should generate a formal CAPA request with defined elements: a clear description of the nonconformity, required root cause analysis, proposed corrective actions with implementation dates, and preventive measures to avoid recurrence.
Track CAPA closure actively rather than waiting for the next scheduled audit. Request evidence of implementation—updated procedures, training records, new test results, or photographs of modified equipment. Define specific re-audit triggers based on finding severity. Critical findings might require verification within 30 days, while lower-priority items can be validated during the next routine audit cycle.
How to Brief Operations So Preventive Controls Actually Reach the Line
Audit findings often identify risks that require immediate action by your own operations team, not just the supplier. When an audit reveals that a supplier’s burst strength testing uses a different standard than your press operators expect, your team needs to adjust their receiving inspection criteria or press settings accordingly.
Create a brief, focused summary of each audit for operations managers and line supervisors. Highlight any changes to incoming material characteristics, new inspection requirements, or supplier-specific handling considerations. This communication transforms audit data into actionable preventive controls that prevent surprises on the production floor.
Audits as the Cheapest Downtime Insurance
An unplanned production stoppage creates cascading costs: lost production time, rush fees to expedite replacement material, overtime for recovery, and potential penalties for missed customer delivery deadlines. These costs typically far exceed the investment in regular supplier audits, whether conducted remotely or on-site.
The true value of audits lies in what doesn’t happen—the quality crisis that never materializes, the port delay that gets prevented, the certificate lapse that’s caught before it impacts shipments. This preventive approach shifts procurement from reactive fire-fighting to proactive risk management, creating more predictable operations and stronger supplier relationships built on verified capability rather than assumptions.
Think of supplier audits as recurring health checks for your supply chain. Just as regular medical checkups catch potential health issues before they become emergencies, structured supplier audits identify process drifts and compliance gaps while they’re still manageable. This evidence-first approach positions procurement and quality teams as proactive leaders who prevent problems rather than simply respond to them.
One Action This Week: Schedule the Next Audit and Send the Checklist
Kraft Paper Supplier Audit—Evidence to Request, Tests to Verify, CAPA Triggers
Pre-Audit Document Requests:
- Current ISO 9001 certificate with scope statement covering your grades
- Active FSC/PEFC certificates if applicable, with registry verification
- Food-contact approvals relevant to your applications
- Three months of lab test summaries for purchased grades, specifying test methods (ISO 287 for moisture, ISO 536 for grammage, ISO 535 or TAPPI T 441 for Cobb, TAPPI T 810 for burst)
- Test reports must include lot-level values with actual test conditions, not only pass/fail indicators
- CAPA log with closed actions from past six months
- Six months of on-time in-full delivery data
- Traceability documentation showing reel/pallet numbers, machine and shift identifiers
- Change notifications for fiber furnish, sizing chemistry, or calendering modifications
On-Site Verification Points:
- Observe manufacturing process at critical control points
- Review calibration records for testing equipment (annual minimum)
- Verify test methods align with purchase order specifications and that samples are conditioned to standard atmosphere before testing
- Confirm testing standards match your specs: ISO 287 for moisture, ISO 536 for grammage, ISO 535 or TAPPI T 441 for Cobb with specified dwell time, TAPPI T 810 for burst
- Check traceability system linking finished product to raw material lots
- Inspect retention sample storage and documentation
- Confirm documented procedures match observed practices
Scoring Framework:
- Critical fail: Expired certificates, no calibrated equipment, broken traceability, vague test reporting without method specification
- Major finding (requires 90-day re-audit): Lab method mismatches, incomplete CAPA closure, significant procedure deviations, unnaturally perfect numbers with no variance
- Minor finding (monitor at next routine audit): Documentation gaps, training record issues, non-critical calibration delays
CAPA Requirements for Findings:
- Root cause analysis documenting why the issue occurred
- Corrective action with specific implementation date
- Preventive measures to avoid recurrence
- Evidence of implementation (updated procedures, training records, test results)
Select one current supplier and schedule their next audit before the end of this month. Send them this checklist two weeks in advance so they can prepare the evidence package. If you’re evaluating new suppliers, use this framework during your pre-qualification process to establish baseline expectations from the start.
Looking to expand your supplier network with pre-vetted partners? Find suppliers or explore our directory of kraft paper manufacturers to connect with mills that can meet your quality and compliance requirements.
Disclaimer: This article provides general educational guidance on supplier audit practices for kraft paper procurement. Specific audit requirements, frequencies, and criteria should be tailored to your organization’s risk tolerance, quality standards, and regulatory obligations. Always consult with your quality assurance and legal teams when establishing supplier audit programs.
Our Editorial Process
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

