📌 Key Takeaways
In pharmaceutical packaging, a low corrugated box price often signals hidden compliance risks that surface as audit findings, shipment holds, and production interruptions.
- Documentation Gaps Cost More Than Price Gaps: Weak traceability, missing change-notification commitments, and certificates that reference misaligned test methods trigger investigations that consume far more resources than any unit-price savings.
- Non-Compliance Is Often Quiet: Problems rarely appear as crushed cartons—they surface as specification mismatches at receiving, unannounced material changes, and lot-to-lot variability that erodes quality system integrity over time.
- Map Failure Modes Before Awarding Business: A Total Cost of Failure framework aligns procurement, QA, and operations on what verification prevents, translating packaging choices into cross-functional risk language.
- Normalize Specifications Before Comparing Quotes: Locking materials, test methods, tolerances, and Incoterms basis transforms quote comparisons from interpretation exercises into genuine value assessments.
- Certificates Alone Cannot Prove Compliance: Valid certificates demonstrate issuing-entity capability, but documentation must link quote, purchase order, shipping paperwork, and the responsible legal entity to survive audit scrutiny.
Qualification discipline protects patient safety; unit price comparisons do not.
Pharmaceutical procurement professionals and packaging engineers managing supplier qualification will gain a practical verification framework here, preparing them for the detailed checklist and FAQ sections that follow.
It is 3:07 PM on a Friday. The quality audit notification for a new regional supplier just failed. Documentation gaps, unverified material changes, and missing traceability records have triggered a hold on three incoming shipments. The production line scheduled for Monday morning now depends on resolving paperwork that should have been verified before the purchase order was ever issued. The cartons look ‘fine.’ The documentation does not. This pattern—where visual inspection passes but verification fails—represents the core challenge in sourcing corrugated boxes.
Think of the pharmaceutical supply chain as an immune system. Its job is to actively reject contaminants—unreliable suppliers, inconsistent materials, documentation gaps—before they compromise the health of the entire operation. When that immune response fails, the consequences spread far beyond a single shipment.
In pharmaceutical supply chains, the unit price of a corrugated shipper represents only a fraction of the Total Cost of Ownership (TCO). What makes a “cheap box” expensive is the risk profile that comes with weak documentation, uncontrolled changes, and inconsistent performance in transit. Those gaps surface as audit findings, shipment holds, rework, expedited replacements, and—at the worst time—production interruptions. The goal isn’t to buy the most expensive box; it’s to qualify a box-and-supplier system that stays stable, traceable, and audit-ready.
Why “Cheap” Is a Dangerous Signal in Regulated Packaging
A quote that undercuts the market often reflects shortcuts in process controls, incoming material verification, or change-management discipline. None of those appear on the quote itself.
A low quote becomes risky when it increases the probability of events such as:
- A receiving or QA hold because the delivered build cannot be linked to the current spec revision
- An unannounced material or process change affecting liner, flute, adhesive, or converting settings
- Higher transit variability that shows up as repacks, expedited replacements, or customer complaints
The false diagnosis trap is confusing price with landed cost, where the latter includes the actuarial risk of non-compliance. A normalized RFQ—one that specifies materials, test methods, tolerances, and Incoterms basis—enables true comparability before price discussions even begin. Procurement teams can submit an RFQ to receive structured, comparable quotes from multiple verified suppliers simultaneously. Without that foundation, comparing quotes means comparing interpretations, not actual value.
Compliance requires adherence to EU Good Distribution Practice guidelines and U.S. cGMP packaging controls under 21 CFR Part 211, Subpart E. Under these frameworks, secondary packaging is a controlled component requiring documented qualification, change control, and traceability. The initial price of a box represents only a fraction of its total risk profile.
What Non-Compliance Looks Like for Corrugated Boxes
Non-compliance is often “quiet.” It shows up as weak traceability, weak change control, or poor lot-to-lot repeatability rather than a dramatic carton collapse.
Documentation gaps include certificates of analysis that reference test methods not aligned with specifications, traceability records that stop at the trader level rather than the manufacturing source, and missing change-notification commitments. Working directly with corrugated box suppliers rather than multi-tier intermediaries can strengthen chain-of-custody verification. When an invoice issuer cannot prove Chain of Custody linkage back to the material origin, compliance certificates on the original quote become meaningless. This pattern represents one of the most common systemic vulnerabilities in Quality Management Systems (QMS): assuming a quote implies compliance capability.
Process gaps appear when incoming inspection reveals variability the supplier never flagged, when storage and handling conditions at the supplier site lack documentation, or when labeling controls fall short of requirements.
Performance gaps manifest as moisture sensitivity that was never characterized, stacking strength variability across production lots, or transit damage rates exceeding what supplier test data predicted. Uncontrolled material or process changes at the supplier level directly increase variance in box performance and receiving nonconformances.
Where the Hidden Costs Actually Show Up
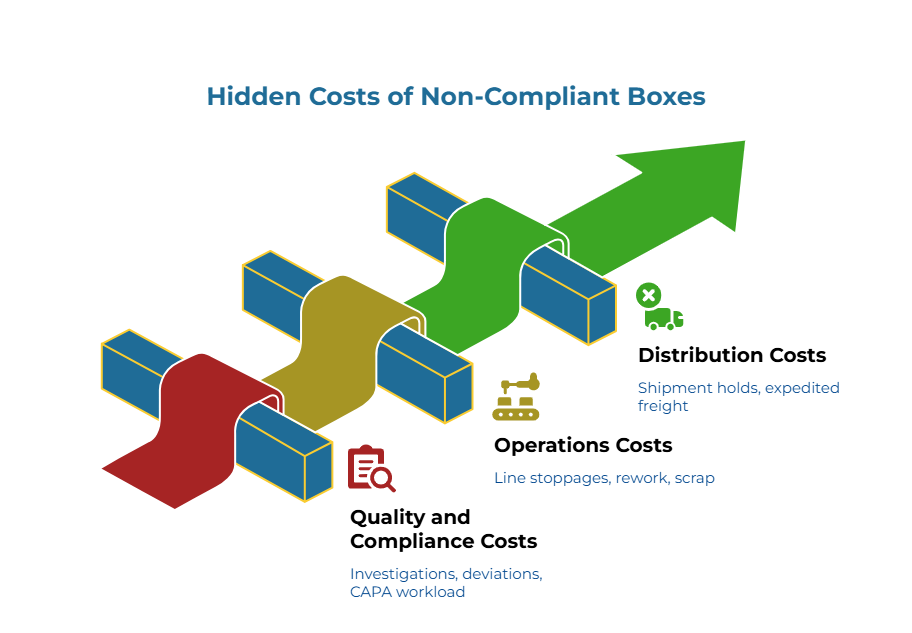
A typical estimated 5–10% variance in unit price is negligible when measured against the cost of a single Critical or Major quality deviation. The biggest cost of a non-compliant box materializes during regulatory inspections and post-market surveillance during audits and investigations—not as visible physical damage. A single undocumented material change can cascade through the quality system for months.
Quality and compliance costs include investigations, deviations, CAPA workload, supplier corrective actions, root-cause analyses, and audit-readiness disruption. These consume QA resources and create downstream delays that rarely appear in unit-price comparisons.
Operations costs emerge as line stoppages, pack-out delays, rework, scrap, emergency reorders, and production schedule disruptions when acceptable shippers are unavailable.
Distribution costs accumulate through shipment holds, expedited freight, repacking, returns, and complaint handling. When boxes fail incoming inspection after arrival, premium freight for replacement orders compounds the original savings into net losses.
These costs distribute across QA, operations, procurement, and logistics—often invisible in unit-price comparisons but very real in operational budgets. A structured sourcing lens that prioritizes repeatability and evidence is outlined in from commodity to assurance: a strategic framework for corrugated box sourcing.
The Total Cost of Failure Framework
Before awarding business based on unit price, map the failure modes that could follow. This worksheet helps align procurement, QA, and operations on what verification prevents.
| Failure Trigger | What Breaks | Downstream Impact | Internal Owner | Preventive Check |
| Supplier changes liner grade without notification | Change control | Deviation investigation, potential product hold | QA + Packaging Engineering | Require change-notification commitment in contract |
| Certificate of analysis references wrong test method | Documentation integrity | Incoming inspection rejection, shipment hold | QA + Receiving | Align test methods in specification before quoting |
| Stacking strength varies significantly across lots | Performance consistency | Transit damage, customer complaints | Packaging Engineering + Logistics | Request multi-lot test reports during qualification |
| Material traceability stops at trader level | Chain of custody | Audit finding, supplier disqualification risk | Procurement + QA | Verify invoice issuer can provide mill-level traceability |
| Moisture content exceeds spec on arrival | Material integrity | Rework, scrap, line delays | Operations + QA | Confirm storage conditions, request COBB test data |
| Spec mismatch at receiving | Conformance | Hold, investigation opened, rescheduling | QA + Operations + Procurement | Approved spec revision history; conformance statement |
This framework surfaces the questions that unit-price analysis alone cannot answer.
A Practical Verification Sequence Before Accepting a Low Quote
Establish specification discipline first. Lock requirements for material grades, test methods (ECT, Mullen, COBB values), dimensional tolerances, and any regulatory-driven specifications. Building a spec-true RFQ creates the foundation for genuine comparison.
Request an evidence pack beyond certificates. Ask for incoming material inspection procedures, recent test reports with method references, traceability documentation samples, and written commitment to notify before any material or process changes.
Define qualification gates before evaluating samples. Run sample quantities through actual receiving processes. Document acceptance criteria in advance—not after results arrive.
Verify the chain of custody when intermediaries exist. Requesting compliance certificates during the RFP process represents standard practice. However, when sourcing through traders or multi-tier supply chains, those certificates may not transfer to the final invoice. Understanding what must appear on paperwork and applying the relay baton test helps verify documentation continuity survives through intermediaries.
More context on why “cheap” often hides avoidable risk is covered in stop buying on price: why ‘cheap’ boxes cost more in the long run.
How to Reduce the Risk of Audit Surprises
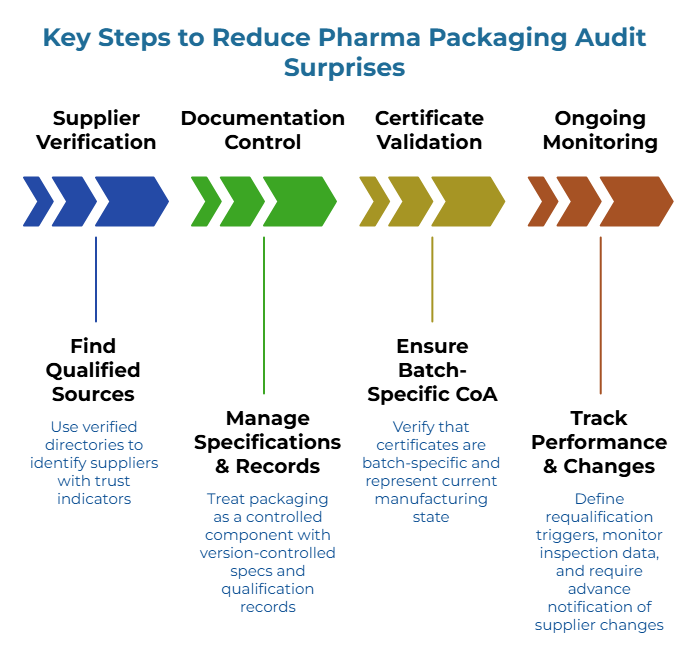
Supplier verification and documentation control reduces audit and shipment-hold risk in pharma logistics. Procurement teams seeking qualified sources can find suppliers through verified directories that include trust indicators such as phone verification and business registration checks. This requires treating corrugated packaging as a controlled component with version-controlled specifications, maintained qualification records, and tracked incoming inspection trends.
Certificates alone can fail when intermediaries exist. A valid certificate proves the issuing entity’s capability—not that the Certificate of Analysis (CoA) is batch-specific or represents the current validated state of the manufacturing line or that claims will appear correctly on invoices. WHO guidance on storage and distribution practices reinforces the importance of documented controls throughout the distribution chain.
Ongoing monitoring matters as much as initial qualification. Define periodic requalification triggers, track incoming inspection data over time, and require advance notification of any supplier-initiated changes.
Quick Checklist: What to Write Into Your Next Corrugated Packaging RFQ
Before sending an RFQ for pharmaceutical-grade corrugated shippers, confirm these elements are specified:
- Dimensions and tolerances; board build; basis weight or GSM; print and registration requirements
- Performance requirements with test methods (ECT or burst, compression or stacking) and conditioning assumptions
- Pack-out and palletization assumptions; storage and handling requirements
- Spec revision control, conformance statement, and traceability identifiers
- Written change-notification commitment covering materials, process, site, and sub-suppliers
- Incoterms basis and named place for quote comparability
- MOQ, lead time, and inclusions affecting total landed cost
A complete RFQ does not guarantee a compliant supplier. An incomplete RFQ guarantees quotes cannot be compared fairly—and suppliers cannot be held accountable later.
Common Questions Procurement Teams Ask
Does a static Certificate of Conformance (CoC) guarantee compliance? While essential, a CoC is a point-in-time snapshot; it does not replace the ‘Golden Thread’ of traceability between the approved specification, what ships, and the responsible legal entity. Documentation must link quotes, PO, shipping paperwork, and the accountable party.
Which tests matter most for corrugated shippers? The right set depends on internal standards and distribution lanes. Many teams combine material indicators such as ECT or burst with compression or stacking strength under defined conditioning. Moisture indicators like COBB values matter for humidity-sensitive routes.
How often should shippers be requalified? Frequency is context dependent. Typical triggers include documented supplier changes, recurring receiving issues, or transit damage signals that exceed baseline expectations.
How can suppliers be compared beyond price? Normalize the RFQ first, then compare the evidence pack: change control commitment, traceability depth, test reporting quality, and repeat-lot consistency. The supplier who documents thoroughly often outperforms the one who quotes lowest.
Price is not value. Value comes from verified comparability and reduced risk. For procurement professionals who see sourcing as stewardship—protecting patient safety through disciplined supplier qualification—the path forward starts with specification discipline, evidence-based verification, and documentation that survives the transaction. The PaperIndex Academy offers additional frameworks for building verification-first sourcing programs across packaging categories.
Ready to discover verified suppliers?Find corrugated box suppliers or submit an RFQ to receive quotes directly.
Disclaimer: This content is for informational and educational purposes only. It does not constitute legal, regulatory, or professional compliance advice. Consult qualified professionals for guidance specific to your organization’s requirements.
Our Editorial Process:
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team:
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

