📌 Key Takeaways
A reactive audit catches failures after they disrupt operations; a structured program prevents them before they reach your facility.
- PDCA Drives Continuous Improvement: The Plan-Do-Check-Act cycle transforms supplier audits from static checklists into living systems that adapt as your business evolves.
- Evidence Beats Assumptions: Cross-checking certificates against public registries (FSC, PEFC, ISO) and comparing supplier test data to your own lab results eliminates disputes and catches problems early.
- Risk-Based Auditing Concentrates Resources: Tier A suppliers warrant quarterly reviews and annual on-site visits, while Tier C suppliers need only annual document checks—focusing effort where supply chain vulnerability is highest.
- Five-Step NCR Protocol Closes Quality Gaps: Contain the material, verify with referee methods, require root-cause CAPA, negotiate commercial resolution, and escalate repeat issues—this sequence prevents recurrence rather than just correcting symptoms.
- 90-Day Rollout Proves the Model: Pilot one grade with one supplier in month one, validate corrections and compare lab data in month two, then lock the framework and add benchmarking suppliers in month three.
Verified capability replaces reactive crisis management. Procurement managers, quality leads, compliance teams, and sourcing heads will find a complete program framework here, preparing them for the detailed implementation guidance that follows.
Building a supplier audit and compliance program isn’t about creating paperwork. It’s about establishing a systematic method to verify that your kraft paper suppliers consistently deliver what they promise—on specification, on certification, and on time. Without a structured program, you’re left with reactive spot-checks that catch problems after they’ve already disrupted your operations.
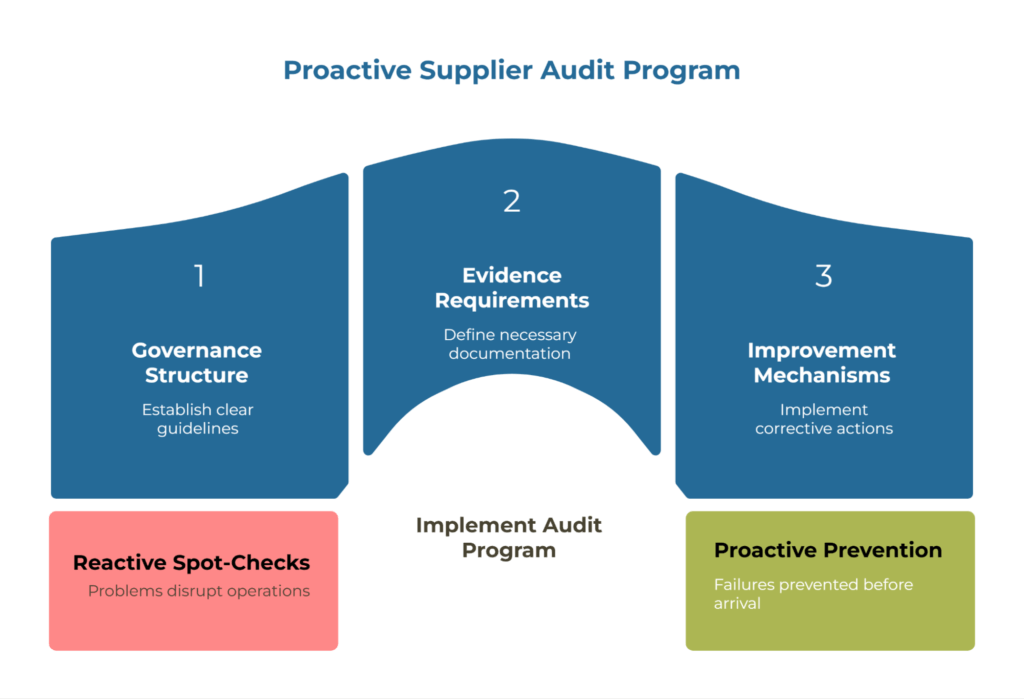
Picture this: A shipment arrives at your receiving dock. The reel tags look fine, the certificates are in order, but when production starts, the Cobb values are off and your converting line backs up for two hours while you scramble to quarantine material and find replacement stock. That’s the moment a properly designed audit program proves its worth—not by catching failures, but by preventing them before they reach your facility.
A well-designed audit program transforms supplier management from a defensive exercise into a strategic advantage. It provides the governance structure, evidence requirements, and improvement mechanisms needed to maintain supply chain integrity while building stronger supplier relationships.
Featured Answer: What This Program Delivers
A Kraft Paper Supplier Audit & Compliance Program is a structured system built on the Plan-Do-Check-Act cycle that defines testable product specifications, verifies a supplier’s management systems and chain-of-custody claims, and monitors performance over time. It combines document reviews, on-site process assessments, statistical sampling, and corrective actions to ensure the kraft paper you purchase consistently matches agreed requirements.
What Is a Supplier Audit & Compliance Program (for Kraft Paper)?
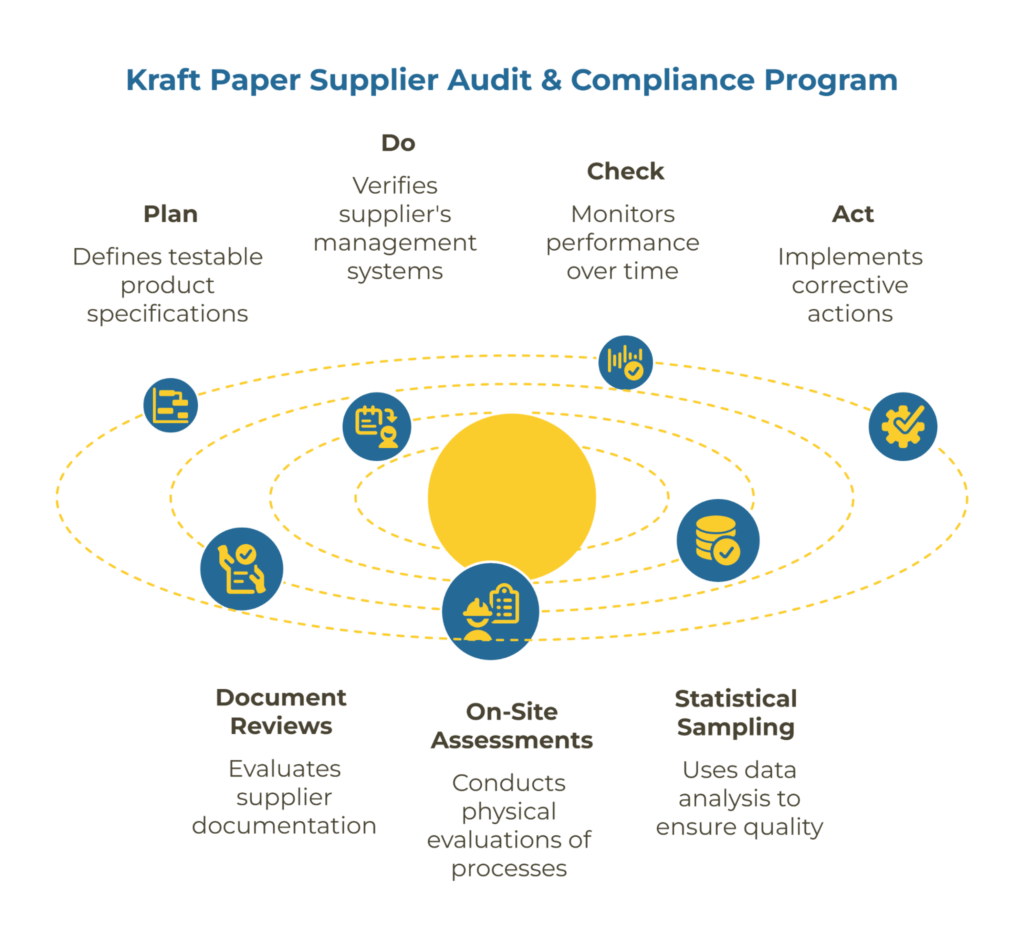
A supplier audit and compliance program is a formalized system for evaluating and monitoring suppliers against your technical, regulatory, and operational requirements. Unlike ad-hoc audits triggered by quality failures, a program approach establishes consistent standards, clear accountability, and continuous improvement processes rooted in the PDCA (Plan-Do-Check-Act) methodology.
The distinction matters because reactive checking creates friction without building capability. When you audit only after problems emerge, you’re managing symptoms rather than addressing root causes. A programmatic approach shifts the focus from crisis response to prevention and partnership.
For kraft paper procurement, this means verifying four interconnected elements: the supplier’s ability to meet your specifications consistently, their chain of custody claims for certified fiber, their quality management systems, and their operational controls for process stability. These elements align with the PDCA cycle—Plan your requirements, Do the verification activities, Check the results against standards, and Act on findings to drive improvement.
The PDCA Foundation
Plan establishes what “good paper” means for your operations. You’ll define testable specifications, determine which certifications matter for your applications, and set clear acceptance criteria. This planning phase also clarifies who makes audit decisions, who performs verifications, and who approves supplier status changes.
Do executes the audit activities: document reviews, on-site mill assessments, sampling protocols, and laboratory testing. This phase generates the evidence that either confirms supplier capability or reveals gaps requiring attention.
Check measures actual results against your planned requirements. You’ll compare mill certificates of analysis to your own test data, verify certificate validity against public registries, and track performance trends across multiple shipments.
Act closes the improvement loop. When gaps appear, you’ll implement corrective actions, adjust your audit frequency based on risk, and refine your requirements as your business needs evolve. This phase prevents your program from becoming a static checklist exercise.
For a practical implementation guide connecting manufacturer evidence to exporter reliability metrics, see the integration playbook.
Program Design: Governance, Roles, and Cadence
Policy and RACI (Who Decides, Who Verifies, Who Approves)
Your audit policy document should answer three questions: What standard must suppliers meet to remain active? What triggers an audit or re-audit? Who has authority to change a supplier’s status based on audit findings?
A RACI matrix clarifies decision rights and prevents the ambiguity that stalls corrective action:
| Activity | Procurement | Quality/QA | Compliance | Operations |
| Define audit scope | C | R | A | I |
| Conduct document review | I | R | A | I |
| Approve new suppliers | C | C | A | I |
| Issue nonconformance reports | I | R | C | I |
| Approve supplier status changes | C | I | A | I |
R = Responsible (does the work), A = Accountable (final decision), C = Consulted (input required), I = Informed (kept updated)
The accountable role for compliance activities typically sits with your regulatory or quality function because they own the interpretation of requirements. Procurement remains accountable for commercial decisions, but compliance determines whether a supplier meets the technical and regulatory bar.
Onboarding vs. Continuous Monitoring
New supplier onboarding requires a more intensive audit covering manufacturing capability, quality systems, and documentation practices. This initial assessment establishes baseline expectations and identifies any immediate disqualifiers before you commit to commercial terms.
Continuous monitoring shifts to a lighter touch, focused on performance trends and periodic re-verification of critical elements. The cadence and depth depend on the supplier’s risk tier and historical performance.
Cadence by Risk Tier (A/B/C Suppliers, Mills vs. Converters)
Risk-based scheduling ensures you invest audit resources where they matter most:
| Supplier Tier | Profile | Document Review | On-Site/Virtual Audit |
| A (Critical) | Strategic volume, specialized capability, or difficult-to-replace | Quarterly | Annually |
| B (Preferred) | Solid performance, moderate volume | Semi-annually | Every 2 years |
| C (Approved) | Backup capacity or lower volume | Annually | As-needed (trigger-based) |
Mills typically receive more frequent audits than converters because they control the base manufacturing process. A converter’s audit focuses on handling, storage, and traceability practices rather than fiber sourcing or chemical management.
Your annual audit plan should map out all scheduled activities, leaving capacity for unplanned audits triggered by quality events, certification expirations, or operational changes at the supplier’s facility.
Audit Scope for Kraft Paper Suppliers: What to Verify
Specification Alignment: GSM, BF/BST, Cobb, Moisture, Tolerances
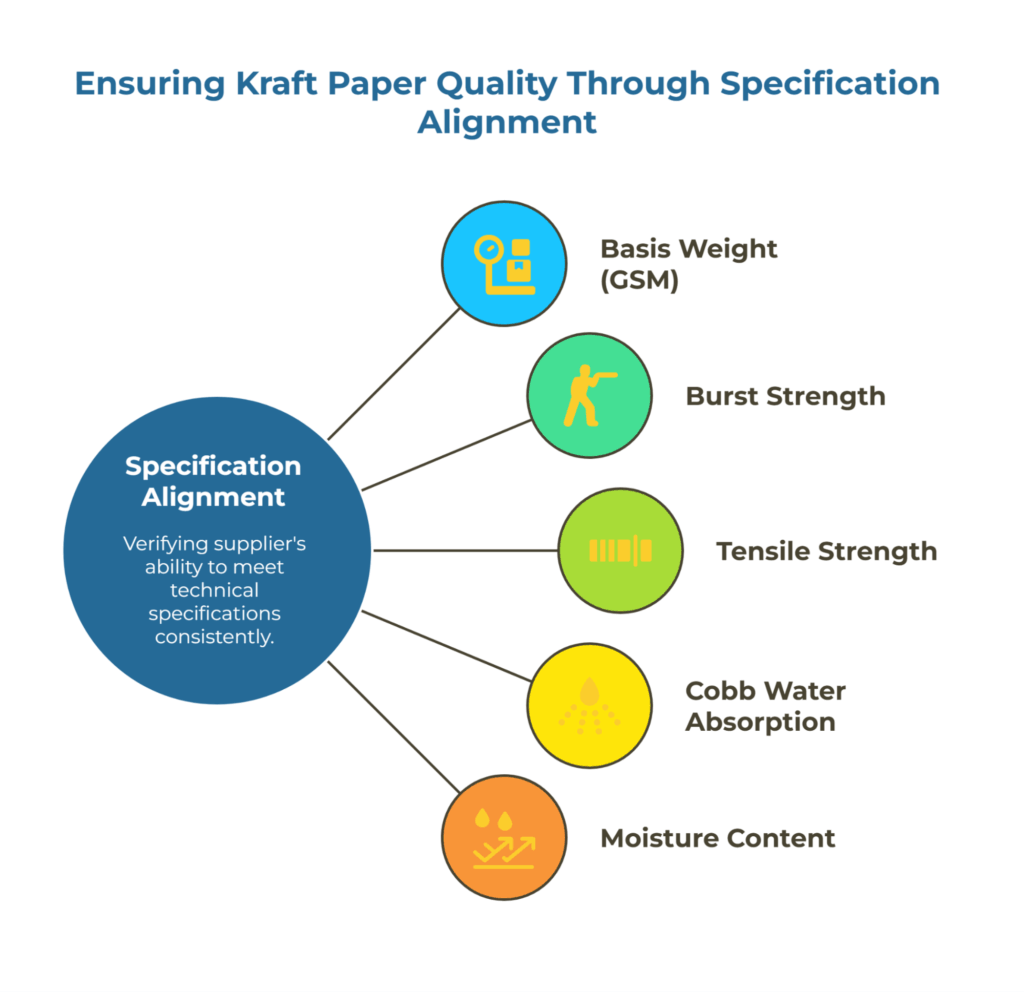
The core of any kraft paper audit is verifying the supplier’s ability to consistently meet your technical specifications. This means reviewing their process capability for the parameters that affect your converting operations or end-use performance.
For basis weight (GSM), check whether the supplier uses ISO 536 methods and maintains calibrated instruments. Ask to see control charts showing their recent production runs—you’re looking for tight distributions within your specified tolerances, not wide spreads that occasionally hit the target.
Burst strength testing should align with ISO 2759 for kraft board applications, while tensile strength follows the ISO 1924 for paper grades. Verify that their test frequency matches your risk profile. A supplier testing once per shift might miss within-batch variation that becomes a problem on your production line.
Cobb water absorption values matter for applications involving moisture exposure. If you’ve specified Cobb 60 or Cobb 1800 limits using ISO 535, confirm the supplier’s test setup follows the standard method and that their reported values include the sizing treatment actually applied to your material.
Moisture content testing per ISO 287 affects both weight accuracy and material performance. Suppliers should demonstrate environmental controls in their storage and packaging areas, not just measure moisture at the end of the line. For detailed specification comparison frameworks, refer to the kraft paper manufacturers capability matrix.
Chain of Custody (e.g., FSC/PEFC) Claims and Continuity
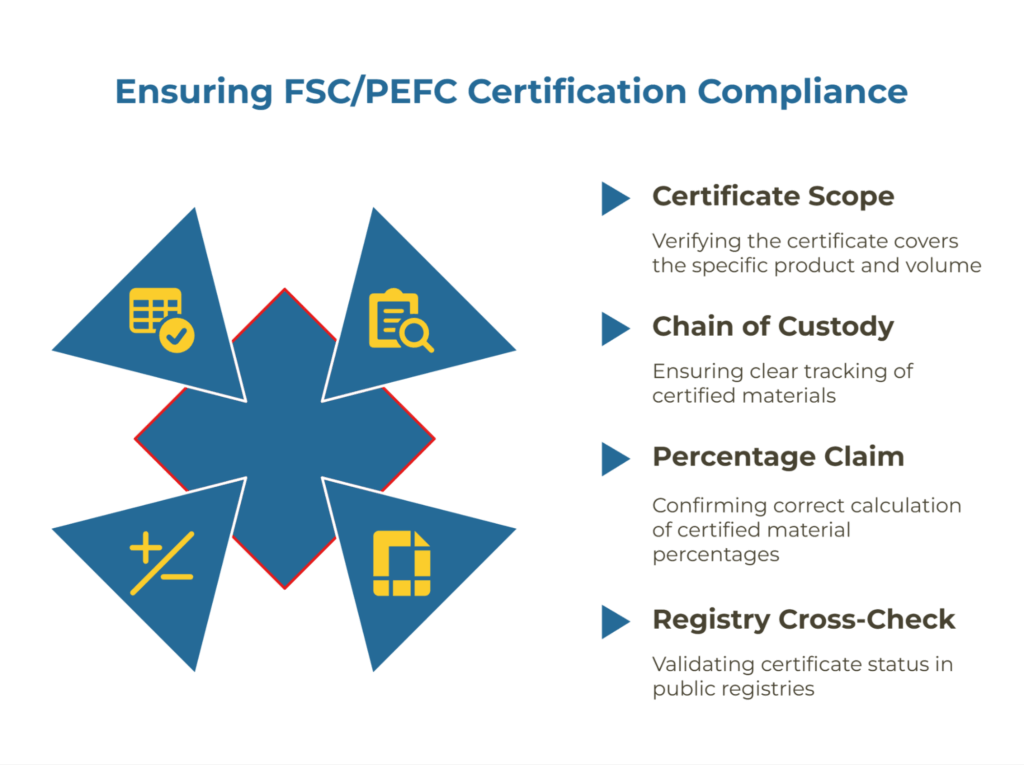
When suppliers claim FSC or PEFC certification, your audit must verify two elements: that their certificate scope covers the specific product and volume you’re purchasing, and that they maintain the documented controls required for chain of custody continuity.
Check the certificate scope statement and any annexes. A supplier with FSC certification for one product line doesn’t automatically extend that certification to all their products. The certificate should explicitly list the material types and the facility where your kraft paper is produced.
Chain of custody documentation should show clear input-output tracking. For FSC Mix or PEFC Certified materials, the supplier’s records must demonstrate they’re maintaining the percentage claim calculation correctly and not mixing certified volumes with non-certified volumes without proper accounting.
Cross-check the certificate number against the FSC database or PEFC registry to confirm active status. Expired or suspended certificates appear in these public registries, providing independent verification of the supplier’s claims.
ISO 9001/14001/22000 Relevance and Site-Scope Matching
ISO certifications provide a baseline indicator of management system maturity, but the audit value comes from verifying site-specific scope and implementation depth.
For ISO 9001 (quality management), confirm the certificate covers the specific facility manufacturing your kraft paper. Multi-site certificates may exclude certain locations or product lines. Review the scope statement carefully rather than assuming wall-to-wall coverage.
ISO 14001 (environmental management) becomes relevant when your procurement includes environmental performance criteria or when you need evidence of chemical management for sensitive applications. The certification itself doesn’t guarantee specific environmental outcomes, but it does signal systematic approach to compliance and improvement.
ISO 22000 (food safety management) applies when your kraft paper enters food contact applications or packaging. However, this certification alone doesn’t replace the specific food contact documentation and declarations required by your end market regulations. Use it as a systems indicator, not a substitute for material compliance evidence. For a comprehensive view of manufacturer verification, see the factory audit for kraft paper manufacturers checklist.
Food-Contact Documentation Patterns (Educational Overview)
Food contact compliance is jurisdiction-specific and requires suppliers to provide declarations aligned with your target markets. In general, you’ll request a declaration of compliance stating the paper meets relevant regulations, a list of substances used in the manufacturing process, and migration test results if your application involves direct food contact or fatty food exposure.
The supplier should be able to trace which raw materials contribute to the final sheet and provide documentation showing those inputs meet applicable purity standards. This traceability becomes especially important when your supply chain includes multiple converters or coating operations.
Your audit should verify the supplier maintains current knowledge of regulatory requirements and has a process for updating formulations or documentation when regulations change. Static declarations that never get revised often indicate a supplier who isn’t actively monitoring their compliance posture.
Operational Controls: Process Stability, Calibration, Packaging
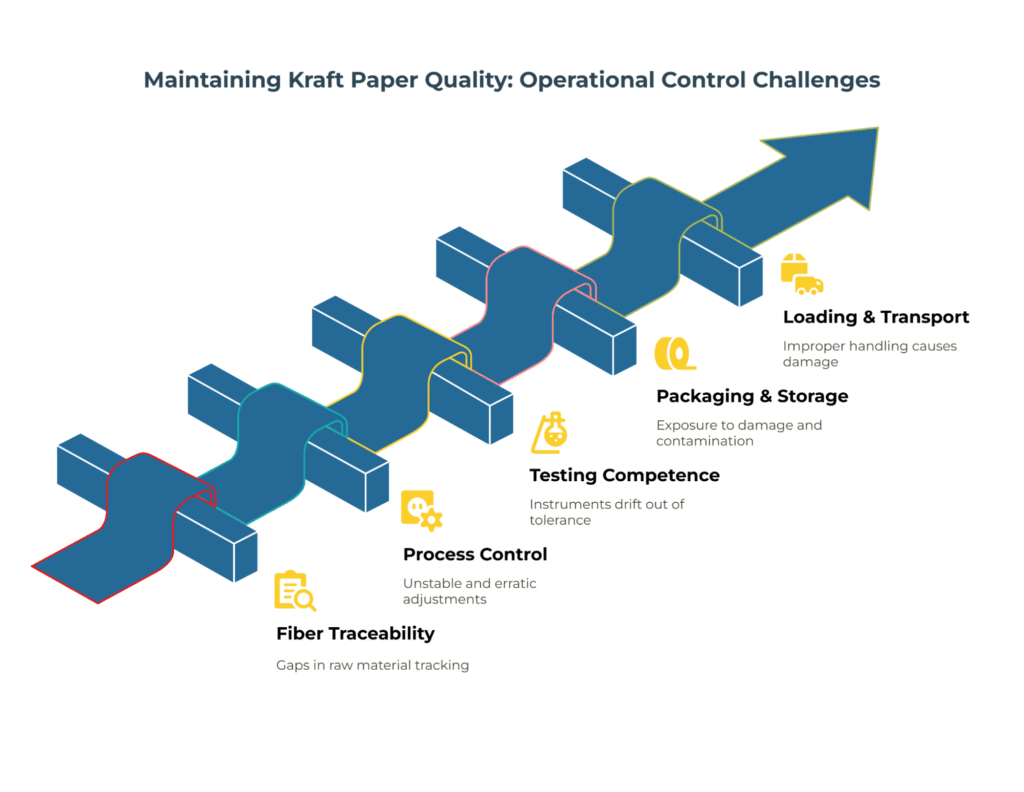
Beyond certificates and test reports, operational controls reveal whether the supplier can maintain performance over time. During on-site audits, focus your assessment on five critical areas:
Fiber inputs and traceability: Verify how batches are identified from pulp receipt through finished reels. Ask to follow a live production lot forward through their system. Strong traceability systems use clear lot coding that connects raw materials to finished goods without gaps or manual interpretation.
Process control capability: Examine their basis-weight control strategy, moisture profiling across the web, size-press or surface treatment settings, and calibration practices for inline gauges. Request control charts for the parameters critical to your application. Stable processes show narrow, consistent distributions; erratic patterns or frequent adjustments suggest underlying capability issues.
Testing laboratory competence: Inspect the instruments used for GSM, Cobb, moisture, and strength testing. Review their internal round-robin results or external calibration records. A well-managed lab maintains traceability to reference standards and documents corrective action when instruments drift out of tolerance.
Packaging and storage practices: Assess how the supplier protects kraft paper from moisture exposure, mechanical damage, and contamination during storage and shipping. Check reel wrap integrity, edge protection quality, humidity monitoring systems, and FIFO (first-in-first-out) stock rotation discipline. Poor handling controls can negate excellent manufacturing.
Loading and transport procedures: Observe reel handling equipment, clamp force settings, and load-securement methods. Improper handling causes telescoping, edge damage, and moisture ingress that only become apparent after the material reaches your facility.
Keep your on-site checklist evidence-oriented. Photographs of gauge readings, anonymized process logs, and sample retain tags provide stronger verification than presentation slides.
Evidence Pack: What to Request, How to Validate, How to Store
Certificates, Scope Annexes, Test Reports, Lot Traceability
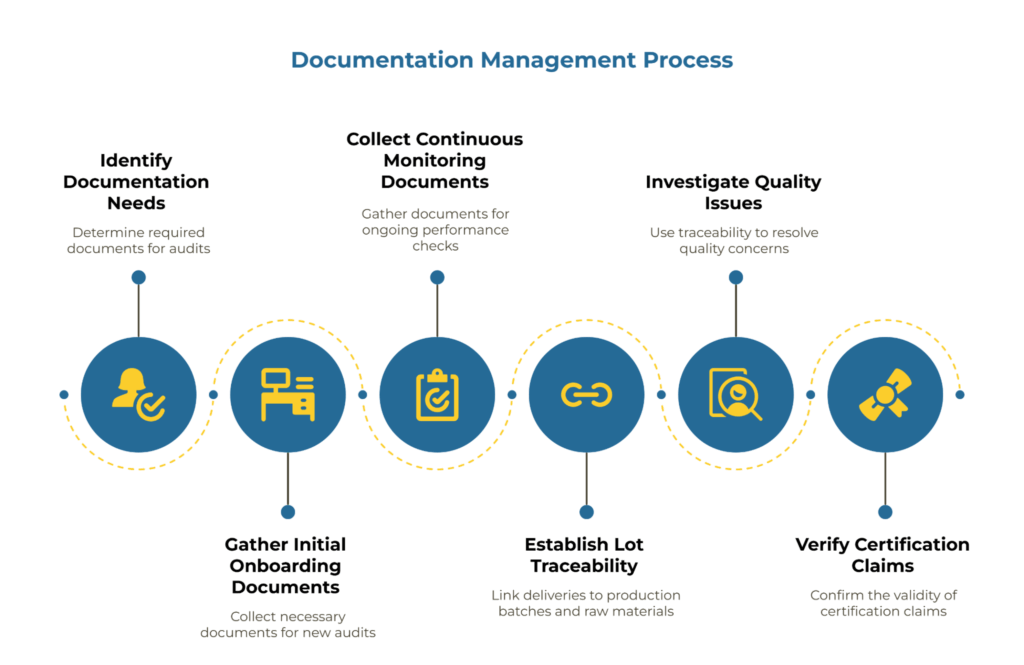
Your evidence pack template should specify exactly what documentation you require for each audit type. For initial onboarding, this typically includes:
- Current quality management system certificates (ISO 9001) with scope annexes
- Chain of custody certificates (FSC/PEFC) with scope annexes if applicable
- Food contact declarations and supporting test reports for relevant applications
- Recent test reports demonstrating specification compliance
- Process capability studies for critical parameters (Cpk values)
- Lot traceability documentation showing material flow from raw inputs to finished goods
For continuous monitoring, streamline requirements to focus on performance evidence: test reports from recent production runs, updated certificates if renewals occurred, and any corrective action reports for quality issues.
Lot traceability records should link a specific delivery to its production batch and, where relevant, back to raw material sources. This connection becomes critical when you need to investigate a quality issue or verify a certification claim.
Registry Cross-Checks, Issuer Validation, Expiry Checks
Never accept a certificate at face value. Cross-check every certification claim against public registries to confirm active status, verify the certificate number matches the registry entry, and check that the scope statement aligns with what the supplier is claiming.
For FSC and PEFC, the official registries are publicly searchable and provide real-time status. Certificates can be suspended or withdrawn for non-compliance, and suppliers don’t always volunteer that information promptly.
ISO certificates should be traceable to an accredited certification body. Check the certificate issuer against accreditation databases to confirm they’re authorized to issue the specific certification being claimed. Unaccredited bodies sometimes issue certificates that look official but lack recognition.
Establish a quarterly verification cycle for all active supplier certificates. This rhythm catches renewals, scope changes, and any suspensions before they become commercial problems. Build a system that flags certificates approaching expiration 90 days in advance, giving you time to request updated documentation before the current certificate lapses.
File Naming, Versioning, and Audit Trail Hygiene
Document control discipline separates functional audit programs from paper-shuffling exercises. Establish a consistent file naming convention that includes the supplier name, document type, issue date, and version number. For example: SupplierName_ISO9001_2024-03-15_v1.pdf
Version control matters when certificates get renewed or when suppliers provide updated test reports. Your system should retain historical versions while clearly marking the current, active document. This version history becomes invaluable during audits or when investigating when a specification change occurred.
Maintain an audit trail showing who received, reviewed, and approved each document. A simple log with columns for document ID, received date, reviewer name, review date, approval status, and notes provides the traceability needed for internal compliance reviews.
Sampling, Inspection, and Acceptance Criteria
When to Sample (Triggers) and Linking to Risk Tier
Receiving inspection intensity should scale with supplier risk tier and material criticality. Tier A suppliers with consistent performance records may require only periodic sampling to verify ongoing conformance. Tier C suppliers or new vendors warrant sample testing on every shipment until they establish a track record.
For small trial orders, test every reel to establish baseline capability. For recurring purchases from proven suppliers, shift to statistical sampling with retained specimens. This risk-based approach concentrates testing resources where uncertainty is highest.
Specific triggers that should prompt immediate sampling include:
- First shipments from a new supplier or a new production line
- Material from a lot not previously tested
- Visual signs of moisture damage, contamination, or handling issues
- Supplier notification of a process change or raw material substitution
- Previous nonconformances on similar materials
Your sampling plan should specify exactly how many sheets or rolls to pull and from which locations in the shipment. Random sampling from different pallet positions reduces the risk of missing localized quality issues.
Acceptance Sampling Concepts (AQL) and Tie-In to CAPA
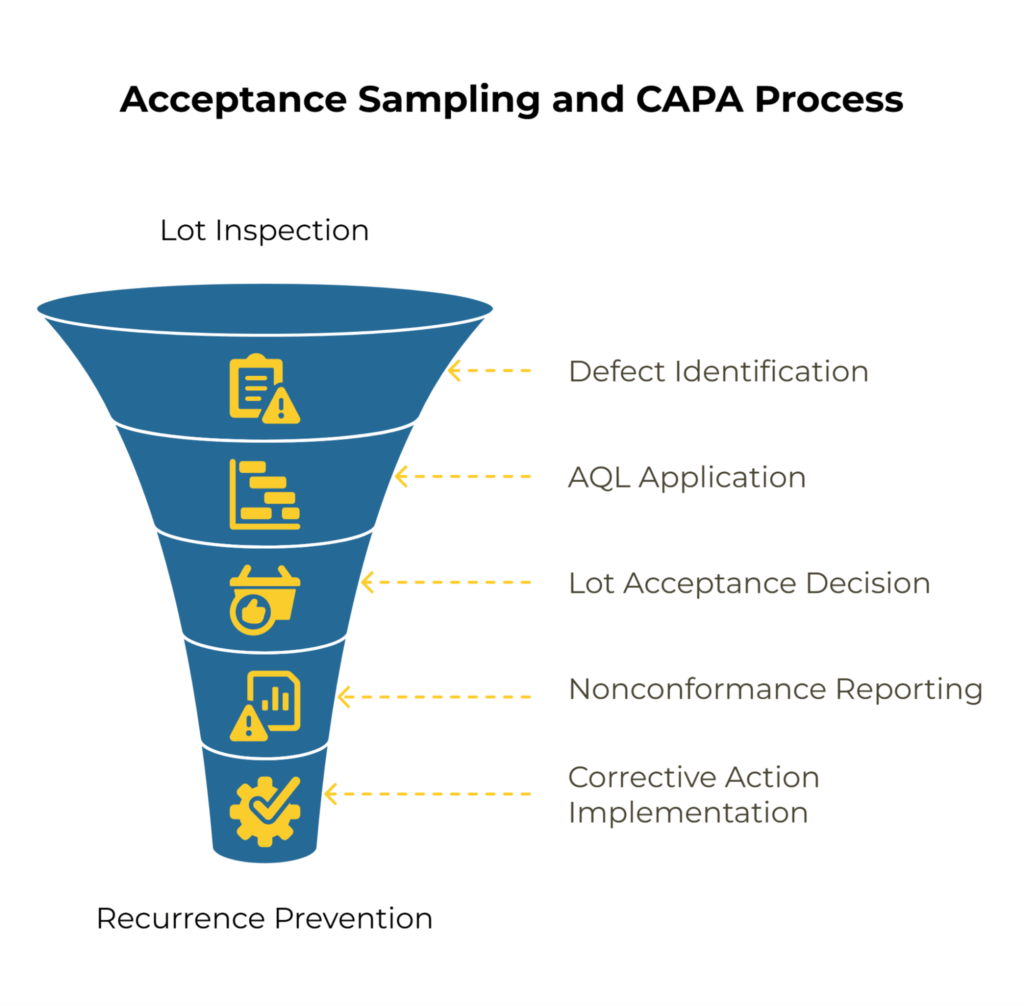
Acceptance Quality Limit (AQL) sampling provides a statistical framework for making lot acceptance decisions based on defect rates. For kraft paper, you might set different AQL levels for different defect types based on their impact on your operations.
Critical defects that make the material unusable (such as severe contamination or major specification misses) typically warrant a 0.0 AQL—meaning zero tolerance. Major defects affecting performance but not rendering the material completely unusable might use an AQL of 1.0 to 2.5. Minor cosmetic issues could accept a higher AQL of 4.0 to 6.5.
The connection to your CAPA (Corrective and Preventive Action) system comes when inspection results fail the acceptance criteria. A rejected lot triggers a nonconformance report that requires the supplier to investigate the root cause and implement corrective measures. Your audit program should track whether these corrective actions actually prevent recurrence.
For detailed guidance on setting AQL levels for kraft paper, see acceptance sampling plans with kraft paper manufacturers.
First-Article vs. Routine Lot Verification
First-article inspection occurs when you’re qualifying a new supplier, validating a specification change, or receiving material from a new production line. This intensive inspection verifies all critical parameters and establishes the baseline for future routine checks.
During first-article inspection, test the full specification suite rather than relying on the supplier’s certificate of analysis alone. Compare your in-house test results to the supplier’s data to identify any methodology differences or calibration gaps that could cause disputes later. This cross-validation also reveals whether the supplier’s test frequency and sampling locations provide adequate process coverage.
Routine lot verification focuses on the parameters most likely to drift or most critical to your application. You’re confirming the supplier maintains control rather than re-proving their entire capability. This streamlined approach makes receiving inspection sustainable while maintaining adequate risk coverage.
Risk Scoring, CAPA, and Re-Audit Windows
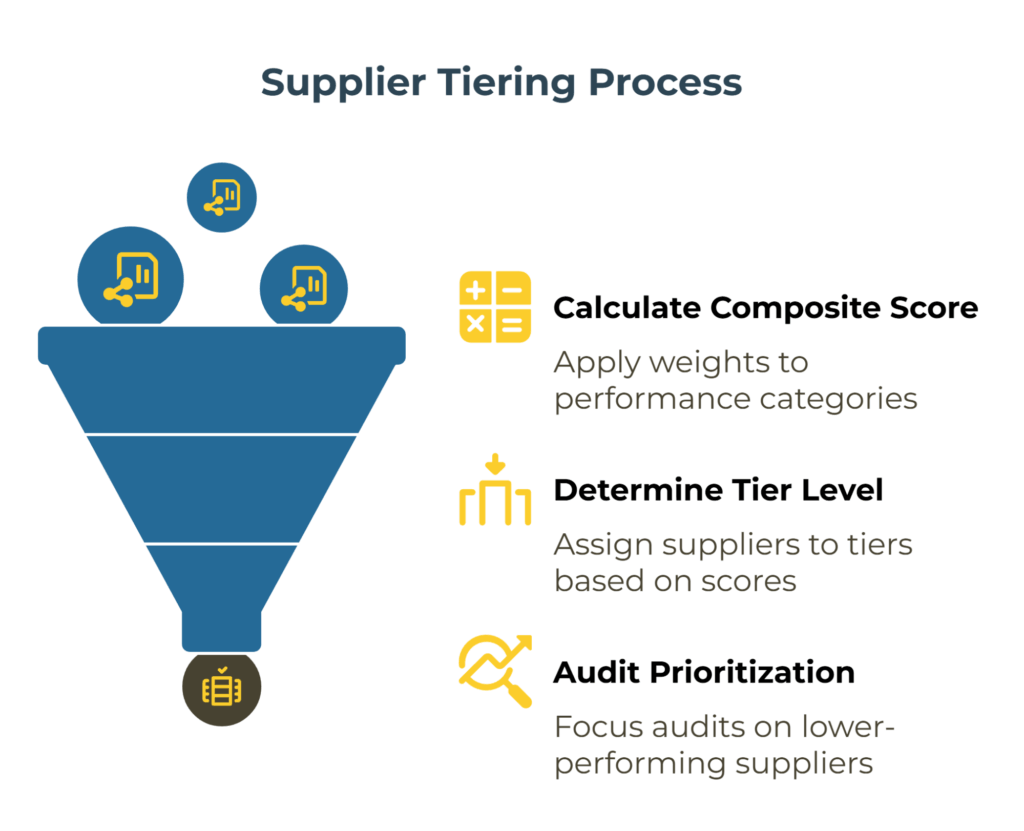
Weighted Scorecard: Documentation Accuracy, Lot Conformance, Delivery Reliability
A numerical risk score provides an objective foundation for supplier tiering and audit prioritization. A typical scorecard weighs multiple performance dimensions:
| Performance Category | Weight | Scoring Criteria (0-5 scale) |
| Documentation accuracy | 25% | Certificate validity, registry match, complete evidence packs |
| Specification conformance | 35% | First-pass lot acceptance rate, test result alignment |
| Delivery reliability | 20% | On-time delivery, complete shipments, lead time consistency |
| Responsiveness to issues | 20% | CAPA closure time, communication quality, proactive disclosure |
Calculate a composite score by multiplying each category score by its weight and summing the results. A perfect score is 5.0. Scores above 4.0 indicate Tier A performance. Scores between 3.0 and 4.0 fall into Tier B. Scores below 3.0 signal Tier C status or potential disqualification.
Update scores quarterly based on rolling performance data. This frequency balances responsiveness to trends against overreacting to individual incidents.
Nonconformance (NCR) Handling and CAPA Effectiveness Checks
When inspection or audit findings identify nonconformances, implement a structured five-step response:
Contain: Immediately quarantine the affected material to prevent mixing with conforming stock. Clear physical segregation and hold tags prevent accidental release to production.
Verify: Re-test the material using your agreed referee method. This step eliminates measurement error as a cause and provides defensible data for any commercial discussions. Where practical, send a retain sample to a mutually agreed third-party laboratory for independent confirmation.
Root cause and CAPA: Require the supplier to provide cause-and-effect analysis and a dated corrective action plan. Effective CAPA addresses root cause, not just immediate symptoms. If a specification miss occurred due to a worn cutting blade, replacing the blade is correction; implementing a preventive maintenance schedule for all cutting equipment is corrective action; adding blade wear monitoring to their process control checks is preventive action.
Commercial resolution: Negotiate appropriate remedies—credits, re-supply at no cost, or material downgrading to a lower-value application. Document the agreement in writing to prevent future disputes about what was decided.
Escalation: Track issue recurrence patterns. Repeated nonconformances on the same parameter within six months indicate the initial corrective action was ineffective and more fundamental intervention is required. This triggers enhanced sampling frequency, potential downgrade to a lower risk tier, or supplier exit if the pattern persists.
Your audit program must include CAPA effectiveness verification. After a supplier implements corrective action, monitor subsequent performance to confirm the issue doesn’t recur. If the same nonconformance reappears within 90 days, the initial corrective action was ineffective and a more fundamental fix is needed.
Re-Audit Criteria and Supplier Status Changes
Define clear triggers that require re-auditing before waiting for the scheduled cycle:
- Multiple NCRs on the same issue within a 6-month period
- Certification expiration or suspension
- Change in ownership, production location, or key personnel
- Major process modifications affecting your materials
- Extended supply pause exceeding 12 months
Re-audit findings determine whether the supplier maintains their current tier status, gets downgraded, or faces disqualification. Document these status change criteria in your audit policy so the decision process remains consistent across different suppliers and audit teams.
Downgrading from Tier A to Tier B triggers increased monitoring frequency and may affect the supplier’s allocation of new business. Movement to Tier C typically restricts them to backup status and requires improvement demonstration before returning to preferred status.
Implementation Roadmap and KPIs
90-Day Rollout: Plan, Do, Check, Act
Launching a supplier audit program across your entire base simultaneously invites chaos. Instead, follow this phased 90-day approach that mirrors the PDCA cycle:
Days 1–30: Plan Phase
Select one or two high-impact kraft paper grades and identify one target supplier for your pilot. During this first month, finalize your testable specification, confirm the supplier’s current certificates (ISO 9001, ISO 14001, FSC/PEFC if applicable), and schedule a focused on-site assessment. Run a controlled trial order with full sampling and retain specimens.
The goal is to validate that your evidence requirements are complete and realistic, test whether your internal review processes function smoothly, and establish baseline performance data. Document the time required for each audit activity to inform resource planning for broader rollout.
Days 31–60: Do and Check Phase
Compare the supplier’s certificates of analysis to your in-house laboratory results. Track on-time delivery performance, physical reel condition at receiving, and the magnitude of any property deltas between supplier data and your tests. This comparison reveals whether methodology differences exist that could cause future disputes.
When gaps appear—whether in documentation completeness, specification conformance, or delivery reliability—work with the supplier to implement targeted corrective actions. Run a small replenishment order to verify the corrections are effective before committing to larger volumes.
Days 61–90: Act and Scale Phase
Lock your final specification, sampling frequency, and acceptance criteria based on pilot learnings. Add a second supplier for the same grade to establish performance benchmarks and reduce single-source dependency. Publish a one-page supplier scorecard template and schedule quarterly business reviews.
This completes your first full PDCA loop and establishes the baseline for continuous improvement. You now have a proven model ready to extend to additional grades and suppliers in subsequent cycles.
Core KPIs: On-Time Quality, Documentation Accuracy, Audit Closure Time, Recurrence of Issues
Measure what matters to your business. The most informative program KPIs include:
First-pass lot acceptance rate tracks the percentage of shipments that pass receiving inspection without rejection or rework. This metric directly reflects supplier capability and your procurement team’s effectiveness at selecting reliable partners. Target rates above 95% for Tier A suppliers.
Documentation accuracy at receiving measures how often suppliers provide complete, valid certificates and test reports with their shipments. Incomplete documentation delays production release and increases your administrative burden. Track this monthly by supplier and set expectations for 98%+ accuracy.
Average CAPA closure time indicates how responsive suppliers are to quality issues and how effectively your team drives issue resolution. Calculate days from NCR issuance to verified corrective action implementation. Best-in-class programs close most CAPAs within 30 days for straightforward issues, 60 days for complex root causes.
Repeat nonconformance rate reveals whether your CAPA process actually drives improvement or just generates paperwork. Track what percentage of NCRs represent issues previously identified and supposedly corrected. Rates above 15% in a rolling 90-day window signal ineffective corrective actions or insufficient verification.
Supplier distribution by risk tier shows whether your base is concentrating risk or diversifying across performance levels. A healthy distribution might show 30-40% Tier A, 50-60% Tier B, and under 10% Tier C. Heavy concentration in Tier C indicates you need to improve supplier selection or development.
Dashboards, Internal Reviews, Supplier Feedback Loops
Build a dashboard that consolidates these KPIs and makes supplier performance trends visible to stakeholders across procurement, quality, and operations. Update the dashboard monthly so it informs sourcing decisions and supplier business reviews.
Quarterly internal program reviews should examine whether audit activities are finding issues before they become operational problems, whether resource allocation matches risk priorities, and whether the program’s requirements remain aligned with your business needs as they evolve.
Supplier feedback loops close the improvement cycle. Share audit findings and performance scores with your suppliers, discuss trends during business reviews, and recognize top performers publicly. This transparency motivates improvement and strengthens partnerships with suppliers who view audits as collaborative quality assurance rather than punitive oversight.
For additional educational resources and industry frameworks, explore the PaperIndex Academy.
Frequently Asked Questions
How often should I audit Tier A versus Tier C suppliers?
Tier A suppliers with critical volume or specialized capabilities warrant annual on-site or virtual audits plus quarterly document reviews. Tier B preferred suppliers can shift to semi-annual document reviews with on-site audits every two years. Tier C approved suppliers need only annual document reviews with on-site audits triggered by performance issues rather than scheduled automatically. This risk-based cadence focuses resources where they protect your supply chain most effectively.
What’s the minimum chain of custody documentation I need to verify FSC or PEFC claims?
You need the supplier’s current FSC or PEFC certificate, the scope statement (often an annex to the certificate), and transaction documentation showing they applied the correct certificate code and volume claims to your purchase orders and invoices. Cross-check the certificate number against the FSC or PEFC registry to confirm active status. For Mix or Certified products, verify the supplier maintains the input-output accounting required to support their percentage claims. The certificate scope must explicitly cover the product type and facility shipping your material.
Does ISO 22000 certification cover all food contact requirements?
No. ISO 22000 demonstrates the supplier operates a food safety management system, but it doesn’t replace the material compliance documentation required by food contact regulations. You still need declarations of compliance specific to your target markets, substance lists for chemicals used in manufacturing, and migration test results if applicable. Use ISO 22000 as evidence of systematic approach, but don’t treat it as a substitute for the regulatory declarations and test data that prove material compliance.
How far back in the supply chain should my audit traceability extend?
For specification conformance, traceability to the production batch is sufficient. For chain of custody claims (FSC/PEFC), you need documented traceability back to certified fiber sources. For food contact applications, traceability should cover all raw materials that become part of the finished sheet or coatings. The practical horizon is typically one to two steps back in the supply chain—from converter to mill, from mill to pulp supplier. Attempting to trace deeper often encounters information barriers that don’t add proportional risk reduction.
What nonconformance threshold should trigger a supplier status downgrade?
Define thresholds in your audit policy based on severity and frequency. As a starting framework: a single critical nonconformance (such as invalid certificates or major specification miss) triggers immediate review for downgrade. Three major nonconformances within six months should mandate downgrade from Tier A to Tier B. Five minor nonconformances in the same period suggest systemic control issues warranting similar action. Failed CAPA effectiveness—where the same issue recurs after supposed correction—should accelerate these timelines.
How long should I retain audit documentation?
Retain audit documentation for the longer of your internal quality system requirements or relevant regulatory retention periods. As a baseline, maintain current audit reports and evidence packs for active suppliers indefinitely. Archive historical audit records for at least three years after a supplier relationship ends. For food contact applications, some jurisdictions require longer retention periods for traceability documentation. Check applicable regulations and err toward longer retention when uncertain. Digital storage makes extended retention practical without significant burden.
Our Editorial Process
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

