📌 Key Takeaways
Supplier approval stalls when procurement and quality operate from different playbooks—shared criteria turn alignment from aspiration into execution.
- Unambiguous Specs Eliminate Disputes: Write specifications so two independent labs reach identical conclusions by naming exact test methods (e.g. ISO 536 for basis weight, ISO 12192 for ring crush) and numerical tolerances—vague language breeds post-delivery conflicts.
- RACI Ownership Prevents Approval Loops: Assign a single Responsible party for each validation point, with clear Consulted and Informed roles documented upfront, so disagreements trigger immediate escalation rather than endless email chains.
- Evidence Quality Trumps Evidence Volume: Demand Certificates of Analysis that include instrument IDs, calibration traceability, and method references matching your specification—accepting incomplete documentation creates ambiguity that surfaces only after the trial order fails.
- Contract Terms Must Enforce Quality Gates: Embed rejection rights, inspection windows, and change-notification clauses directly into purchase orders so quality commitments carry legal weight rather than relying on supplier goodwill.
- Sealed Retention Samples Settle Disputes: Store representative samples from each lot under controlled conditions with documented seal numbers and photos—this evidence transforms opinion-based conflicts into fact-based resolutions.
Alignment beats assumption when both teams reference the same thresholds, ownership, and evidence requirements.
Procurement managers, quality assurance professionals, and operations leads will find the structured framework here, setting the stage for the detailed checklist and implementation guidance that follows.
Now that you’ve collected Pre-Qualification Questionnaires, reviewed audit findings, and narrowed your shortlist, the final approval step remains: getting procurement and quality to agree on the same decision criteria.
This is where many supplier evaluations stall. Procurement focuses on lead times and commercial terms. Quality demands specific test thresholds and compliance documentation. Operations wants clarity on who owns the handoff. Without a shared framework, what should be a straightforward approval becomes a cycle of back-and-forth emails, missed sign-offs, and delayed trial orders.
The solution isn’t more meetings. It’s a single-page checklist both teams can reference during the final review—one that specifies what evidence is required, who validates it, and what constitutes a pass or fail. This article delivers that tool: a practical, executive-friendly approval framework designed to move a candidate kraft paper supplier from “shortlist” to “approved for trial order” with clear ownership and unambiguous thresholds.
The Short Checklist:
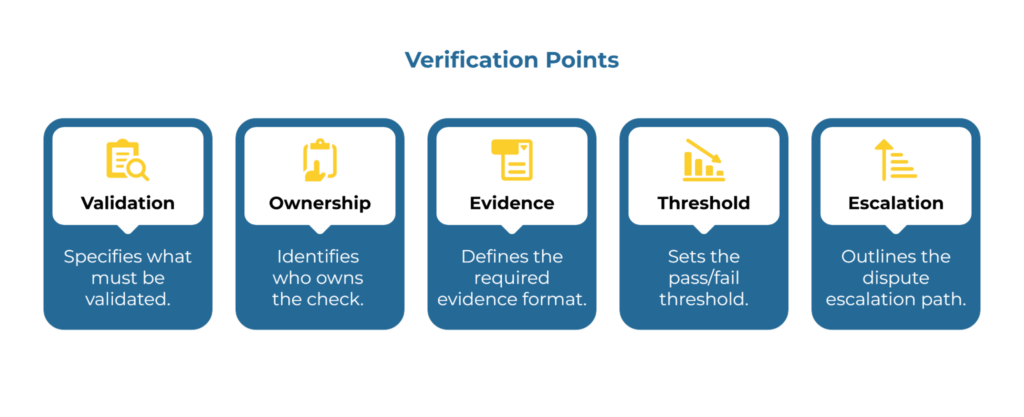
Shared acceptance criteria reduce rework and disputes. The table below provides ten verification points your procurement and quality teams can use to finalize supplier approval. Each item specifies what must be validated, who owns the check, what evidence format is required, the pass/fail threshold, and the escalation path if disputes arise.
Write your specification so that two independent labs would reach the same conclusion. For each property you care about—basis weight, tensile strength, burst resistance, or water absorptiveness—name the exact test method and state the tolerance numerically. This approach produces defensible results and eliminates the “close enough” disputes that delay approvals.
Procurement × Quality: Supplier Approval Checklist
| # | What to Verify | Owner (RACI) | Evidence Required | Pass/Fail Threshold | Escalation |
| 1 | ISO 9001 or equivalent certification current and in scope | Quality (R), Procurement (C) | Certificate scan + surveillance audit date | Valid through trial period + 6 months | Procurement Director |
| 2 | Lab accreditation (ISO/IEC 17025) for claimed test methods | Quality (R) | Accreditation certificate with scope listing | All requested tests in scope | Quality Manager |
| 3 | Basis weight tolerance meets specification (±3% of nominal) | Quality (R) | Lab report per ISO 536 from 3 production rolls sampled per ISO 186 | ≤3% deviation from target GSM | Quality Manager |
| 4 | Tensile strength (MD and CD) meets minimum thresholds | Quality (R) | Lab report per ISO 1924-2; MD/CD data separate; instrument ID or calibration traceability included | MD ≥4.5 kN/m; CD ≥2.0 kN/m (adjust per spec) | Quality Manager |
| 5 | Short-span compression (SCT) or ring crush meets application requirements | Quality (R) | Lab report per ISO 9895 or ISO 12192 with conditioning data and method reference | SCT-CD ≥2.2 kN/m or RCT ≥value per end-use | Quality Manager |
| 6 | Chain-of-custody certification (FSC/PEFC) matches PQQ claim | Quality (R), Procurement (C) | Current CoC certificate + transaction confirmation | Certificate valid; covers claimed fiber sources | Procurement Director |
| 7 | Sample conditioning protocol documented and ISO-compliant | Quality (R) | Supplier test plan citing ISO 187 (23±1°C, 50±2% RH); conditioning duration ≥24 hours | Protocol meets ISO 187; samples equilibrated before testing | Quality Manager |
| 8 | CAPA process documented with turnaround commitments | Quality (R), Ops (C) | Supplier CAPA SOP + historical closure times | Median closure ≤30 days for non-conformances | Operations Manager |
| 9 | Lead time and MOQ align with production planning | Procurement (R), Ops (C) | Confirmed lead time + MOQ in supplier’s quote | Lead time ≤8 weeks; MOQ ≤2 production runs | Procurement Director |
| 10 | Audit report (remote or on-site) addresses identified risks | Quality (R), Procurement (I) | Audit report with CAR status + closure evidence | All high-risk CARs closed or mitigated before trial | Quality Manager |
RACI Key: R = Responsible (performs the check), A = Accountable (final sign-off), C = Consulted (provides input), I = Informed (notified of outcome)
Sampling and Acceptance Rules: Define your lot boundary, sampling plan, and accept/reject logic before the first shipment arrives. Use ISO 186 to establish representative sample selection procedures. Where attribute-based acceptance applies, reference ISO 2859-1 for your AQL plan. Document sample size, acceptance number, and retest conditions in writing—this prevents post-delivery disputes about what constitutes a valid test.
This checklist transforms abstract alignment into concrete accountability. Each row answers three questions: What needs verification? Who confirms it? What evidence settles the matter? By anchoring decisions to measurable thresholds rather than subjective judgment, the framework accelerates safe supplier approval while reducing the risk of misaligned expectations during the trial order phase.
PQQ responses, sample test results, and documented CAPA protocols de-risk the first trial order. When both teams reference the same ten checkpoints, the approval discussion shifts from defending departmental concerns to validating shared evidence.
Key Terminology
Cross-functional teams work more efficiently when everyone speaks the same technical language. These definitions provide quick reference for the properties and methods most commonly debated during approval meetings:
GSM (Grammage): Mass per unit area, typically expressed in grams per square meter. This foundation property influences strength characteristics and material yield.
RCT (Ring Crush Test): Compressive strength measurement for paper and linerboard, often used as a performance proxy in corrugated packaging applications. Test method: ISO 12192.
Burst Strength (Mullen): Resistance to rupture under applied pressure. Common in legacy specifications for kraft liners and sack papers. Test method: ISO 2759.
Tensile Strength: Pulling resistance measured under controlled elongation conditions. Reported separately for machine direction (MD) and cross direction (CD). Test method: ISO 1924-2 or TAPPI T 494.
Cobb60: Water absorptiveness measured over a 60-second interval. Critical for moisture-sensitive applications. Test method: ISO 535.
AQL (Acceptance Quality Limit): The maximum percent defective (or maximum number of defects per hundred units) that can be considered acceptable as a process average under an attribute sampling plan. Reference: ISO 2859-1.
How to Use the 1-Page Checklist (and Avoid Common Pitfalls)
A checklist is only effective if teams follow a consistent review process. The three-step usage framework below ensures your approval meetings stay focused and productive.
Step 1: Pre-Read (48 hours before the meeting)

Distribute the completed checklist to all stakeholders at least two business days in advance. Each owner listed in the RACI column should validate their assigned items and flag any red or amber statuses. This pre-work prevents the approval meeting from becoming a first-look data review session. If quality identifies a failed threshold on item 4 (tensile strength below minimum), procurement can request clarification or a retest from the supplier before the joint review.
When validating lab reports during pre-read, cross-check the Certificate of Analysis numbers against your stated tolerances and methods rather than assuming “meets spec” based on supplier claims. If the COA shows burst strength via Mullen test but your specification calls for ring crush, that mismatch must be resolved before approval—the tests measure different failure modes and are not interchangeable.
Step 2: Joint Review (30-minute meeting)
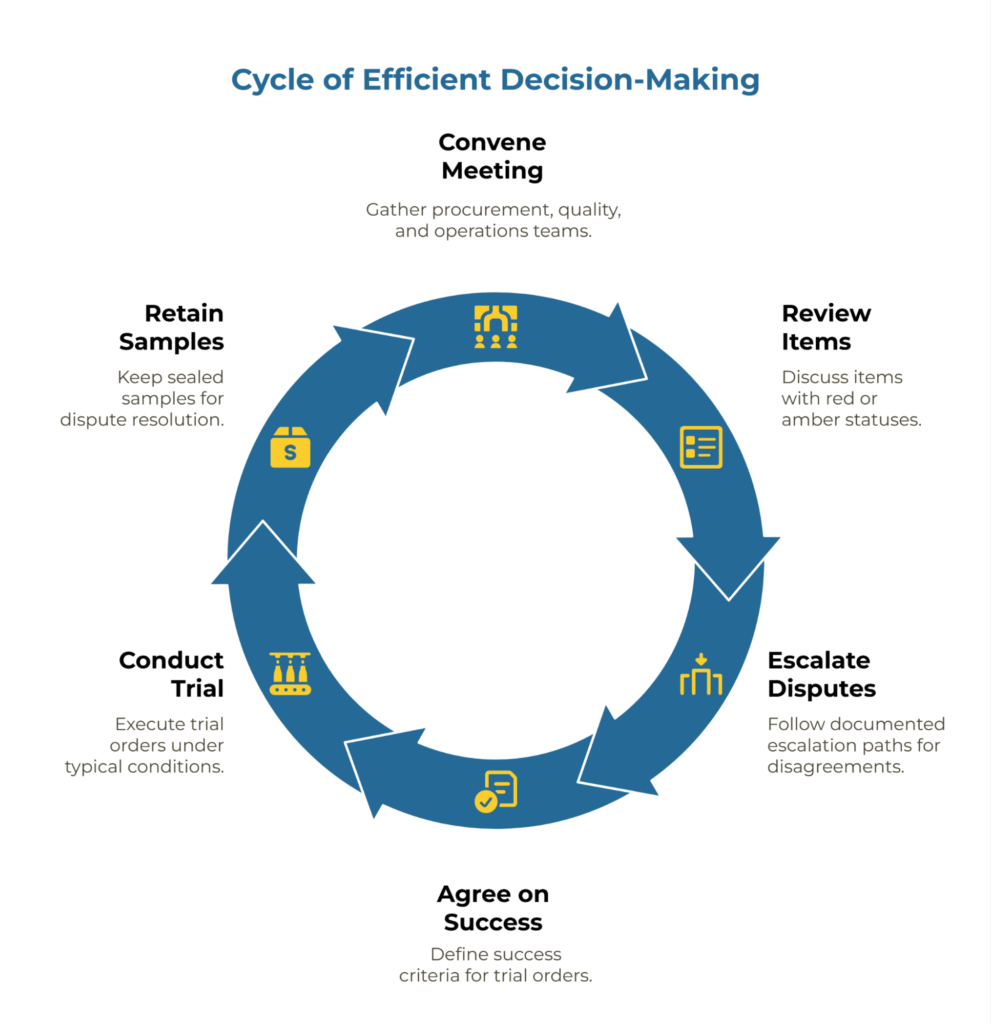
Convene procurement, quality, and operations in a single session. Walk through each of the ten items in sequence, focusing only on red or amber statuses. Green items require no discussion beyond confirmation. For disputed items, apply the documented escalation path immediately rather than deferring to “offline follow-up.” If procurement and quality disagree on whether a remote audit (item 10) sufficiently addresses identified risks, escalate to the Procurement Director per the checklist’s escalation column. This discipline prevents approval delays caused by unresolved judgment calls.
Size your initial trial order to allow a representative line trial across typical production shifts and operating conditions, while limiting financial exposure if rework becomes necessary. Agree upfront on what constitutes trial success—using the same methods, tolerances, and acceptance logic documented in the checklist—and keep sealed retention samples for evidence in case disputes arise later.
Step 3: Decision (same meeting)
After reviewing all items, apply a three-tier approval framework: Approve (all items green; issue PO for trial order), Probation (1-2 amber items with clear mitigation plan; conditional approval with 30-day CAR), or Reject (any red item; supplier returns to shortlist pending corrective action). Document the decision with date, final status, and accountable signatories on the checklist itself. This creates an audit trail for future supplier reviews and ensures both teams share ownership of the outcome.
When Alignment Succeeds—and When It Fails
Scenario 1: Method Precision
Success case: Your specification calls for ring crush strength per ISO 12192 with a defined minimum threshold. The supplier’s Certificate of Analysis references the same test method and instrument calibration traceability. Your receiving lab runs the same ISO 12192 protocol, and results align within measurement uncertainty. Approval proceeds without debate.
Failure case: The supplier reports only Mullen burst strength values. Your internal team assumes “close enough” based on historical correlation tables and approves the supplier. During production scale-up, downstream corrugated board shows inadequate top-to-bottom compression performance because burst and ring crush measure fundamentally different structural properties. The assumption that one predicts the other fails under your specific board design and humidity conditions.
Scenario 2: Sampling Discipline
Success case: Your receiving team draws samples per ISO 186 protocol, logs container seal numbers and reel positions, photographs the packaging condition, and stores retention samples in a controlled environment. When a quality issue surfaces three weeks later, you have defensible evidence showing the material arrived in specification and was handled correctly.
Failure case: A busy receiving dock spot-checks one reel “because we’re behind schedule.” A container with moisture intrusion goes unnoticed because no systematic moisture readings were taken. Customer complaints arrive, but you lack the documented evidence to demonstrate whether the problem originated at the mill, during transit, or in your facility. The dispute becomes opinion versus opinion, and resolution drags for months.
Scenario 3: Contract Enforcement
Success case: Your purchase order ties acceptance to the written specification and checklist criteria, includes a 10-business-day inspection window, and requires 30-day advance notification before any change to fiber sources, sizing chemistry, or machine configuration. When the supplier proposes a cost-saving fiber substitution, you receive formal notification, run qualification tests, and approve or reject the change based on documented evidence.
Failure case: The PO states only “subject to inspection” with no method references, tolerance windows, or notification requirements. The supplier makes an undisclosed fiber blend change. Performance degrades slightly but noticeably. When you attempt rejection, the supplier argues the material “meets industry norms.” Without contractual definitions of acceptance, the dispute escalates to legal arbitration.
Email Template for Internal Circulation
Standardizing how approval decisions are communicated across departments ensures all stakeholders receive the same information in a consistent format. The template below can be copied into your email client and customized with supplier-specific details.
Subject: [DECISION REQUIRED] Kraft Paper Supplier Approval – [Supplier Name]
To: [Procurement Director], [Quality Manager], [Operations Manager]
CC: [Sustainability Officer if applicable]
We have completed the joint procurement-quality review for [Supplier Name] using our 10-point approval checklist. Below is the decision summary:
Decision: [Approve / Probation / Reject]
Summary:
• Risk Assessment: [Brief statement of key risks identified during PQQ/audit phase, e.g., “Remote audit flagged inconsistent conditioning protocols; supplier submitted corrective SOP.”]
• Evidence Validated: [List 2-3 critical passed items, e.g., “ISO 9001 valid through Q2 2026; ISO/IEC 17025 scope covers all requested tests; FSC CoC confirmed for claimed fiber sources.”]
• Outstanding Items: [List any amber/red items and mitigation plan, e.g., “SCT results 0.1 kN/m below threshold; supplier resampling from different production lot; retest due by 2026.”]
• Trial Order Parameters: [If approved, specify PO value/volume, e.g., “Issue trial PO for 50 MT at agreed commercial terms; first shipment expected week of 2026.”]
• Compliance Checkpoint: [If probation, specify review trigger, e.g., “CAR #2024-Q3-05 closure required within 30 days; quality to verify before releasing second PO.”]
Next Steps:
- [Owner name] to [specific action, e.g., “issue trial PO by EOD Thursday”]
- [Owner name] to [specific action, e.g., “schedule 30-day progress review for probation items”]
- [Owner name] to [specific action, e.g., “update supplier database with approval status and trial order notes”]
Owners & Dates:
• Procurement Lead: [Name] – PO issuance by 2026
• Quality Lead: [Name] – Incoming inspection protocol confirmed by 2026
• Operations Lead: [Name] – Production scheduling updated by 2026
For questions or escalations, refer to the attached checklist or contact [Accountable Party].
This email structure ensures that anyone reading the thread—including stakeholders who weren’t in the approval meeting—can immediately understand the decision, the supporting evidence, and their specific responsibilities. The five-bullet summary format aligns with how executives scan emails: decision up front, evidence in the middle, action items at the end.
FAQ: When Procurement and Quality Disagree
Even with shared criteria, interpretation differences occasionally arise. The scenarios below address the most common points of friction and provide evidence-based resolution paths.
Problem: Procurement wants to proceed with a supplier whose lab results are borderline, but quality insists on retesting before approval.
Response: The checklist’s pass/fail thresholds exist to prevent subjective judgment calls. If tensile strength results (item 4) fall within 5% of the minimum threshold, quality’s request for resampling from a different production lot is justified. Kraft paper properties vary by reel position and production timing; a single marginal result doesn’t confirm consistent performance.
Evidence: ISO 2859-1 (sampling procedures for inspection by attributes) recommends increasing sample size when initial results approach specification limits. Approving a supplier based on borderline data creates risk during scale-up. The cost of a retest—typically $200–400 and 5–7 business days—is negligible compared to the cost of rejecting an entire shipment post-delivery due to out-of-spec material.
Problem: Quality demands an on-site audit before approval, but procurement argues the supplier’s remote audit and ISO 9001 certification are sufficient.
Response: The decision framework for item 10 depends on the identified risk level during PQQ review. If the PQQ template: minimum evidence required from kraft paper suppliers flagged high-risk areas—such as inadequate traceability controls for mixed fiber sources or recent quality hold incidents—an on-site audit is non-negotiable before trial order approval.
Evidence: Remote audits effectively verify documentation and management systems but cannot assess physical process controls, material handling practices, or operator competency. For suppliers in regions with inconsistent regulatory oversight or those producing specialty grades requiring tight tolerances, on-site verification reduces the probability of post-approval surprises. ISO 19011 (auditing management systems) explicitly notes that remote audits have limitations for evaluating site-specific risks.
Problem: Procurement prioritizes a supplier with faster lead times, but quality flags that their CAPA process (item 8) has historically slow closure times.
Response: Lead time advantages become irrelevant if the supplier cannot resolve non-conformances quickly. CAPA turnaround is a leading indicator of operational discipline. A supplier with a 45-day median CAPA closure time may deliver faster initially, but quality issues will create downstream delays that negate any scheduling benefits.
Evidence: Reference the supplier’s designing CAPA workflows for kraft paper suppliers: from finding to fixing documentation. If their process lacks defined escalation triggers or doesn’t include root cause analysis for recurring issues, the risk of extended downtime during the trial phase is high. Procurement’s lead time priority should be conditional on CAPA performance meeting the ≤30-day threshold.
Problem: The supplier passed all checklist items, but operations raises concerns about MOQ misalignment after the approval meeting.
Response: This represents a process failure, not a checklist failure. Item 9 explicitly requires operations to be consulted during the MOQ validation step. If Operations wasn’t included in the joint review or didn’t flag the concern during Step 2, the solution is procedural: reconfirm the supplier’s flexibility on order quantities before issuing the trial PO, or escalate to the Procurement Director if the MOQ genuinely cannot support production planning.
Evidence: MOQ disputes discovered post-approval typically signal incomplete stakeholder engagement during the pre-read phase. The RACI model in the checklist exists to prevent this scenario—ensuring operations validates practical feasibility before the decision is finalized, not after.
Connecting Approval to Ongoing Assurance
Supplier approval isn’t a one-time gate; it’s the entry point to a continuous monitoring relationship. Once you issue the trial purchase order, the evidence-gathering process shifts from pre-qualification validation to in-process verification and post-delivery audit.
The frameworks you used to finalize approval—documented thresholds, clear ownership, and evidence-based escalation—should extend into your kraft paper supplier onboarding checklist: from PQQ to first trial order and ongoing supplier management protocols. The same RACI logic that clarified approval responsibilities applies to incoming inspection hand-offs, CAPA tracking during the trial period, and periodic re-audits after the supplier achieves approved status.
During receiving inspection, protect quality through systematic logistics controls. Verify packaging integrity, check for edge protectors and moisture barriers, and confirm desiccant presence in containers. Take moisture readings at arrival and photograph wrapping condition and seal numbers. Where water sensitivity matters for your application, measure Cobb absorptiveness per ISO 535 on representative samples. This documentation preserves evidence if disputes arise and provides the baseline for accepting or rejecting shipments under your contract terms.
Your commercial terms should reinforce quality rather than undermine it. Build acceptance criteria directly into the purchase order: explicit right of rejection tied to your written specification, a defined inspection window (typically 10 business days), mandatory retention sampling, and a change-notification clause for critical parameters like fiber sources, sizing chemistry, or machine configuration. Without these contractual teeth, quality erodes into “we’ll check later” informality, and disputes become difficult to resolve.
Quality should schedule the first post-delivery audit no later than 90 days after the initial shipment. This timing allows sufficient production data to assess consistency but catches issues before the supplier is deeply embedded in your supply chain. Use the checklist: what to require in a kraft paper supplier audit report to ensure your audit scope aligns with the risk areas identified during approval.For lab test validation during incoming inspection, your quality team should reference how to interpret lab test reports for kraft paper to verify that supplier-provided certificates match your internal acceptance criteria. The same conditioning protocols (ISO 187), method IDs (ISO 536, 1924-2, 9895), and MD/CD balance requirements from the approval checklist must carry forward into your receiving inspection procedures.
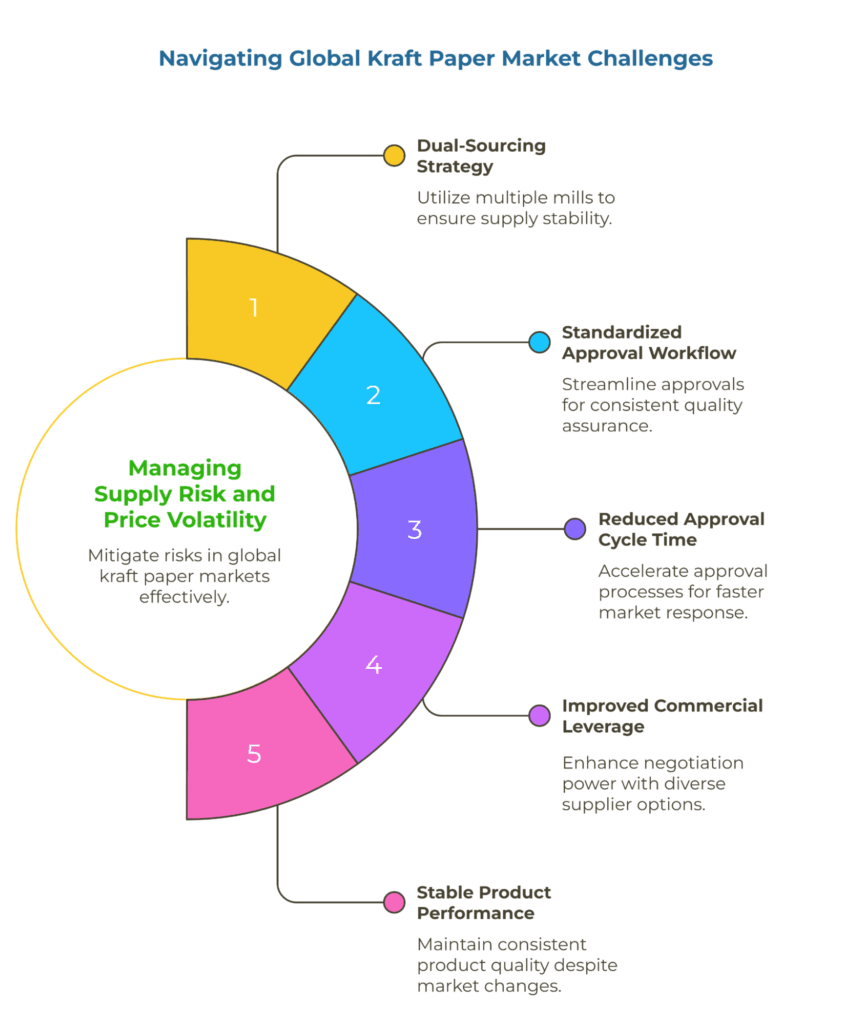
To manage supply risk and price volatility across global kraft paper markets, maintain at least two qualified mills across different geographic origins. Standardize your PQQ and approval workflow so quality evidence remains comparable—especially when working with smaller exporters who may not volunteer detailed method documentation unless explicitly requested. This dual-sourcing approach reduces approval cycle time, improves commercial leverage, and keeps product performance stable through market fluctuations and regional supply disruptions.
Procurement × Quality alignment accelerates safe supplier approval. When both functions operate from shared acceptance criteria rather than parallel judgment calls, decisions move faster and disputes decrease. The one-page checklist, email template, and three-step review process transform abstract collaboration into concrete accountability—ensuring your next kraft paper supplier moves from shortlist to approved status with clarity, confidence, and minimal friction.
References:
[1] ISO 9001:2015, Quality management systems—Requirements, Section 8.4 (Control of externally provided processes, products and services).
[2] ISO 19011:2018, Guidelines for auditing management systems, Section 6 (Performing audit activities).
Our Editorial Process
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

