📌 Key Takeaways
Three documents—kept together and easy to find—turn inspection stress into a calm folder handoff.
- Inspections Test Retrieval, Not Quality: Most packaging inspection failures happen because operators can’t locate paperwork quickly, not because containers are unsafe.
- Three Documents Cover Everything: A letter of guarantee, supporting compliance evidence, and traceability proof form a complete inspection-ready set for each packaging item.
- Match Documents to What’s Actually On-Site: Compliance paperwork only works when it clearly links to the specific products currently in your kitchen through invoices and lot codes.
- One Folder Per Product Keeps It Simple: Separate folders with numbered subfolders (01 Guarantee, 02 Evidence, 03 Traceability) make retrieval automatic under pressure.
- Request Documentation Before You Order: Ask suppliers for all three documents during onboarding—waiting until an inspector asks creates gaps you can’t close quickly.
Organized paperwork means confident inspections.
Restaurant owners and food service operators managing takeout packaging will gain a clear system for inspection readiness, preparing them for the detailed filing guide that follows.
~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~
Food-contact compliance during an inspection often hinges on three specific documents. While local enforcement varies, maintaining a structured filing system ensures immediate, verifiable proof of packaging safety
Three specific documents and a structured filing system replace the frantic search with immediate, verifiable proof. Local requirements and enforcement vary, so treat this as a readiness framework rather than a guarantee—but operators who can produce basic compliance paperwork quickly rarely find themselves in prolonged conversations with inspectors.
Most inspection stress around takeout packaging isn’t about proving the paper itself is good. It’s about producing basic compliance paperwork quickly when asked. Inspectors aren’t testing your containers on the spot. They want to see that you can back up a simple claim: this packaging is appropriate for contact with food.
What Inspectors Are Really Looking For
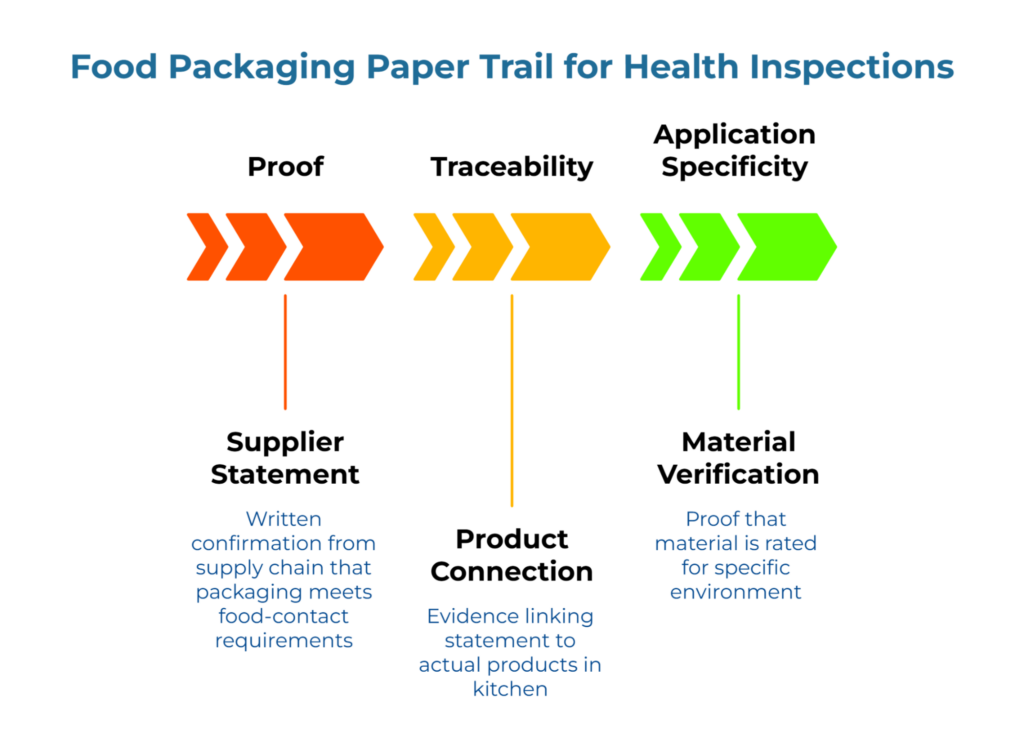
Health inspectors aren’t packaging engineers. They respond to paperwork—traceable, dated proof—more than verbal assurances. When an inspector asks about your food-contact packaging, they’re checking three things.
Proof: A written statement from someone in your supply chain confirming the packaging meets food-contact requirements. Not your word. Documentation.
Traceability: Evidence connecting that statement to the actual products in your kitchen right now. The inspector’s unspoken question is straightforward: Is this the same product the document refers to?
Application Specificity: Verification that the material is rated for your specific environment (e.g., thermal resistance for hot liquids vs. grease barriers for fried foods). Generic ‘food safe’ labels are insufficient for specialized use cases, which is why auditing suppliers for FDA and ISEGA safety through scope-matched verification gates matters.
When operators fail this part of an inspection, it’s rarely because they bought bad packaging. The common pattern: paperwork can’t be located, or what’s available doesn’t clearly match what’s on the shelf.
The 3-Document Inspection-Ready Checklist
- Letter of Guarantee or compliance statement
- Supporting compliance evidence
- Traceability proof (invoice, lot code, COA, or shipping reference)
What Each Document Proves
| Document | What It Proves | Who Provides It | Fast Validation Check | Where to File It |
| Letter of Guarantee / compliance statement | The packaging is intended for food contact | Supplier or manufacturer | Product name matches your SKU; intended use is stated clearly | Front of the product folder |
| Supporting compliance evidence | The claim has a regulatory or testing basis | Supplier, manufacturer, or third-party lab | Document is dated; references a standard or test relevant to your use | Behind the letter of Guarantee |
| Traceability proof | The documents apply to what’s actually on-site | Your records (invoice, shipping docs) | SKU/product name matches; lot code aligns if provided | Behind the compliance evidence |
Letter of Guarantee or Compliance Statement
This is your anchor document. A letter of Guarantee is a written assurance from your supplier stating that the packaging is suitable for its intended food-contact use. While specific requirements vary, this practice is grounded in federal regulations (such as 21 CFR 7.13) which establish the framework for compliance guarantees in the supply chain.
What it should include:
- The specific product identified (not just “paper containers” but the actual SKU or product name)
- The intended use stated clearly (food contact, and ideally the type—hot, cold, greasy, dry)
- A clear compliance assurance referencing the regulatory basis
What to check: Does the letter name your specific product? Does it address your actual use case? A letter covering “dry food contact” doesn’t help if you’re using the container for hot, greasy fried chicken.
What to do if it’s vague: Ask your supplier for a revised version that names the product clearly and specifies the intended use. A generic statement won’t satisfy an inspector who wants to know whether these specific clamshells are appropriate for hot, greasy food.
Where to file it: Front of the folder for that product. This is the first document you’ll pull.
Supporting Compliance Evidence
The letter of guarantee makes a claim. Supporting evidence backs it up.
This might be a reference to a regulatory standard like 21 CFR 176.170 for paper and paperboard in contact with aqueous and fatty foods, a migration test report, or documentation showing how the supplier verified compliance. This documentation aligns with the FDA Food Code requirements (specifically Section 4-102.11), which mandate that single-service and single-use articles be clean and safe for contact with food.
You don’t need to understand every technical detail. You need documentation that’s relevant to your use case and recent enough to be credible.
What counts as supporting evidence:
- A reference to the applicable FDA regulation or food-contact standard
- Test reports from the manufacturer or a third-party lab
- A declaration of compliance citing specific regulatory provisions
What “current” means: Documentation practices vary, but evidence dated within the past two to three years is generally reasonable for unchanged products. If your supplier changes formulations, new documentation should follow.
Red flags to watch for:
- Documents with no date
- Generic “food safe” language without any regulatory reference
- Test reports that don’t match the product name on your invoice
- Documentation for dry goods when your use involves hot or greasy food—a mismatch that underscores why understanding GSM and Kit Levels in food packaging paper matters for specification accuracy
Where to file it: Behind the letter of guarantee in the same product folder.
Traceability Proof
This is the “paper trail” that ties everything together.
Consider a compliance manager at a busy quick-service restaurant. The inspector is asking about clamshells, and she has a letter of guarantee—but it references a legacy SKU or a formulation that may have been updated eighteen months ago. If the documentation doesn’t explicitly match the current product specs, the paper trail is broken—a failure mode that the integrated safety model addresses by linking each spec line to verification gates. Twenty minutes of digging through emails, and the inspection is still stalled.
Traceability proof prevents this. It connects your compliance documents to the specific products currently in your kitchen.
What to file:
- Invoice showing the product name, SKU, and supplier
- Lot or batch code from the packaging (if provided)
- Certificate of analysis or COA (if provided)
- Shipping reference that ties the shipment to the documentation
The specific format matters less than the connection it establishes. When an inspector asks whether your documentation applies to the containers on your shelf, you can point to the invoice and say: this document covers this product, and here’s the shipment record showing this is what I received.
Where to file it: Behind the compliance evidence, completing the three-document set for each product.
How to Assemble Your Compliance Binder
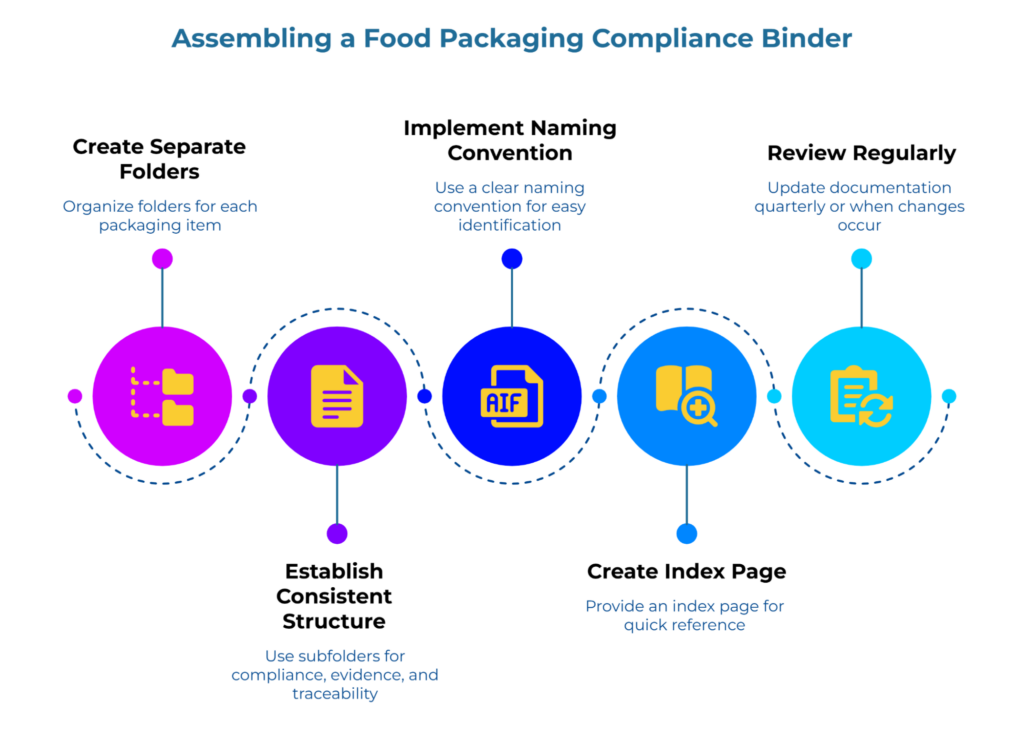
Make this painfully simple. You’re not building an archive—you’re creating a folder you can hand to an inspector in under sixty seconds.
One folder per packaging item. Separate folders for cups, clamshells, paper bags, wraps—whatever you use regularly. If you have multiple sizes of the same product from the same supplier with the same documentation, they can share a folder. Different products or different suppliers get their own.
A consistent internal structure. Inside each product folder, keep three subfolders in order: 01 Guarantee/Compliance, 02 Supporting Evidence, 03 Traceability. The numbering keeps them sorted correctly and makes retrieval automatic—you’re always reaching for the same sequence.
A simple naming convention. Product — Supplier — Doc Type — Date. “Clamshell_8oz — AcmePackaging — Guarantee — Jan2025” tells you exactly what you’re looking at without opening the file.
An index page. One sheet at the front of the binder listing each packaging item and confirming its three documents are present. This is your cheat sheet when the inspector asks a question—and your reminder when something needs updating.
A light update rhythm. Review quarterly, or whenever you change suppliers or SKUs. The change trigger matters more than the calendar—compliance documentation can become outdated if suppliers change formulations or your use case shifts, so respond to actual changes rather than relying solely on scheduled reviews. For guidance on how documentation gaps develop over time, this guide on how compliance decay happens in food-grade packaging paper explains what to watch for.
A Simple Script to Request Documents from Suppliers
Most suppliers will provide this documentation when asked clearly. Here’s a template:
Subject: Documentation Request for [Insert Product Name or SKU]
Hi [Supplier Contact],
I’m organizing our food-contact compliance records and would appreciate your help gathering documentation for [specific product name/SKU].
Could you please provide:
- A letter of Guarantee or compliance statement confirming this product is suitable for food contact (ideally specifying the intended use, such as hot or greasy foods if applicable)
- Supporting documentation that backs up the compliance claim (regulatory reference, test report, or similar)
- Confirmation of how shipments are tied to this documentation (lot codes, COA references, or invoice linkage)
This helps us stay organized for health inspections. Thank you for your help.
Keep the tone cooperative. You’re asking for standard documentation, not accusing anyone of wrongdoing.
If a supplier can’t or won’t provide basic documentation, that’s worth noting. Suppliers who struggle to produce compliance evidence often have gaps elsewhere in their quality systems—a pattern explored in depth through the seven questions to ask a new supplier that scammers can’t answer. For more on evaluating supplier documentation practices, this guide on how to verify food contact compliance beyond certificates covers key questions to ask, and this analysis of why food packaging paper suppliers fail compliance verification explains typical failure modes.
Frequently Asked Questions
Do health inspectors actually ask for packaging paperwork?
They can. Requirements vary by jurisdiction and inspector focus, but food-contact packaging falls under the broader category of food safety documentation. Being prepared costs you nothing; being caught unprepared costs time and creates stress during an already demanding moment.
What is a letter of Guarantee for food-contact packaging?
A letter of Guarantee is a written statement from a supplier or manufacturer assuring that a product is suitable for its intended food-contact use. It typically identifies the product, states the intended use, and references the regulatory basis for the compliance claim. The FDA recognizes these letters as part of the food-contact compliance chain.
What if my supplier only says “food safe” but won’t share documentation?
Ask for clarification. A reputable supplier should be able to point to a regulatory basis for their claim—a specific FDA regulation, a test report, or a declaration referencing applicable standards. For detailed guidance on distinguishing verified claims from vague assurances, see why ‘food safe’ is a meaningless label and the seven questions that separate credible suppliers from those who cannot substantiate their claims. If they can’t provide anything beyond verbal assurance, consider whether that’s a level of risk you want to carry.
How often should I update this documentation?
Review quarterly or whenever you change products, suppliers, or SKUs. Documentation that was valid a year ago may not reflect current formulations. If you’re using the same product from the same supplier and nothing has changed, a simple confirmation that the documentation remains current is reasonable.
What should I keep for imported packaging?
The same three documents apply, but traceability becomes more important. Make sure you can connect your compliance documentation to the specific shipment you received, especially if goods pass through multiple intermediaries—a challenge addressed in detail by the zero-trust sourcing model for verifying overseas suppliers. Additional documentation—such as Third-Party Lab Migration Tests (SGS/Intertek)—is critical for imported goods to verify they meet U.S. safety standards. For guidance on matching test conditions to actual use, see the workflow for navigating Specific Migration Limits across global compliance frameworks.
What if I have a new supplier and no documentation yet?
Request it before placing your first order, or at minimum before the product goes into use. Don’t wait until an inspector asks. Building the habit of requesting documentation as part of supplier onboarding prevents gaps from developing.
What if lot codes are missing from my packaging?
Use what you have. An invoice with the product name, date, and supplier still provides traceability even without a lot code. Note the gap and ask your supplier whether lot codes can be provided on future shipments. Some products simply don’t carry them, and that’s not automatically disqualifying—but the more connection points you can document, the stronger your paper trail.
What if I have multiple similar SKUs?
Create separate folders for each distinct product. A 12-oz clamshell and an 8-oz clamshell may come from the same supplier, but they could have different compliance documentation. When in doubt, keep them separate.
From Scrambling to Ready
The inspection moment doesn’t have to be stressful.
Three documents. One binder. A sixty-second retrieval time. That’s the difference between scrolling through emails behind the counter and handing over a folder without breaking stride.
When your compliance paperwork is organized, you’re not just prepared for inspectors—you’re running the kind of operation where nothing catches you off guard. Having these records centralized ensures the inspection remains a routine verification rather than a high-stakes audit.
For more practical guides on food-contact compliance and supplier verification, explore the PaperIndex Academy. If you’re ready to connect with food packaging paper suppliers who can provide proper documentation, the marketplace offers a starting point for that search.
Disclaimer:
This information is for educational purposes. Requirements vary by jurisdiction; consult with a local environmental health specialist to ensure full compliance.
Our Editorial Process:
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team:
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

