📌 Key Takeaways
“Food safe” on a label proves nothing—only named standards with current, lot-linked documents verify safety.
- Labels Are Marketing, Not Proof: The words “food safe” have no legal definition and can appear on packaging without any testing or certification behind them.
- Three Documents Prove Compliance: A Declaration of Compliance naming standards, a migration test matching your use conditions, and lot traceability linking papers to your shipment.
- Ask for Standards by Name: Suppliers who answer “which standard?” with vague phrases like “food grade” or “meets all standards” likely cannot prove compliance.
- Old Certificates Can Be Worthless: When suppliers change ingredients, coatings, or factories, past test results no longer apply to current shipments.
- Documentation Is a Purchase Requirement: Make compliance papers part of every quote request—not something you scramble for after an inspector asks.
Current documents beat comforting labels every time.
Procurement managers and food service operators sourcing food packaging paper will gain a practical question framework here, preparing them for the detailed supplier verification guidance that follows.
~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~
A shipment arrives at the receiving dock. The carton says “food safe.” No certificate attached. No standard reference. Just two words printed on cardboard.
Is this actually safe to use?
That doubt is justified. A receipt is not a warranty; documentation is the warranty. The phrase “food safe” sounds like a promise, but it proves nothing on its own. Without a named standard and current documentation tied to the material, those two words are marketing—not compliance.
Food contact safety standards are the specific regulatory benchmarks (such as FDA 21 CFR in the U.S. or BfR Recommendations in Europe) that define which materials are chemically safe for direct contact with consumable goods—a topic explored comprehensively in food-grade certification standards for delivery packaging bags. Specifically, compliance with these regulations acts as the legal authorization for the material’s use. Inspection readiness requires verifying specific regulatory citations or third-party certificates (such as those from ISEGA) against every invoice to maintain a verifiable chain of custody.
The Myth: What “Food Safe” Implies
Most buyers assume “food safe” means a product has been tested, certified, and approved for contact with consumable goods. The phrase implies third-party validation—that the material has undergone rigorous toxicology and migration testing to ensure consumer safety.
That assumption is wrong. “Food safe” has no standardized legal definition. A supplier can print it on packaging without referencing any specific standard, test, or certification body. Different markets require entirely different proof: US food service typically references FDA 21 CFR Part 176.170 for paper and paperboard. In Europe, while the Framework Regulation (EC) No 1935/2004 applies generally, there is no harmonized EU-wide legislation specifically for paper; therefore, industry typically relies on national standards, most notably Germany’s BfR Recommendation XXXVI, to demonstrate compliance.
Without naming the specific standard, “food safe” tells you nothing about which rules the material actually meets.
The Reality: What Actually Proves Safety
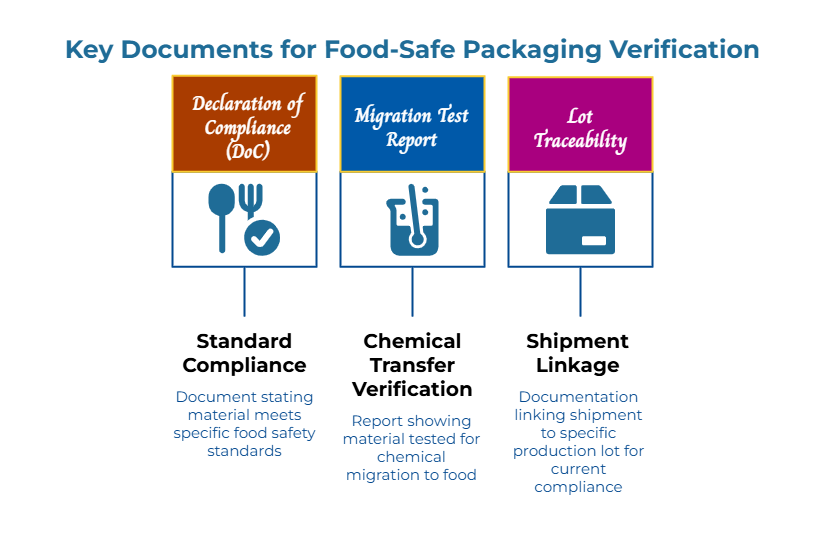
Verification requires three documents—no more, no less.
A Declaration of Compliance (DoC) that explicitly names the standard(s) the material meets and describes the scope: what product grades, what food types, what use conditions (time and temperature).
A migration test report showing the material was tested under conditions matching your intended use. Migration testing verifies that chemicals don’t transfer to food at unsafe levels. The report should specify the test method, food simulants used, and time/temperature conditions.
Lot traceability linking documentation to the actual shipment—a principle that applies equally to verifying supplier capability beyond price considerations. A certificate from eighteen months ago doesn’t prove today’s shipment is compliant—especially if the supplier changed raw materials, coatings, or inks since then.
The critical distinction is “current” and “lot-linked.” A certificate is a snapshot in time. If a supplier changes their formulation, the old certificate becomes invalid even though they may still present it as proof—a phenomenon examined in depth in why food-safe labels fail: how compliance decay happens in food-grade packaging paper. When the lot code on your shipment doesn’t match the lot referenced in the documentation, you don’t have proof. You have hope.
Consider a common scenario: a restaurant switches to a cheaper wrapper labeled “food safe” and later cannot produce a migration test or DoC during inspection prep. The lesson is stark—make documentation a purchase requirement, not a post-incident scramble.
The Questions to Ask (And What Good Answers Look Like)
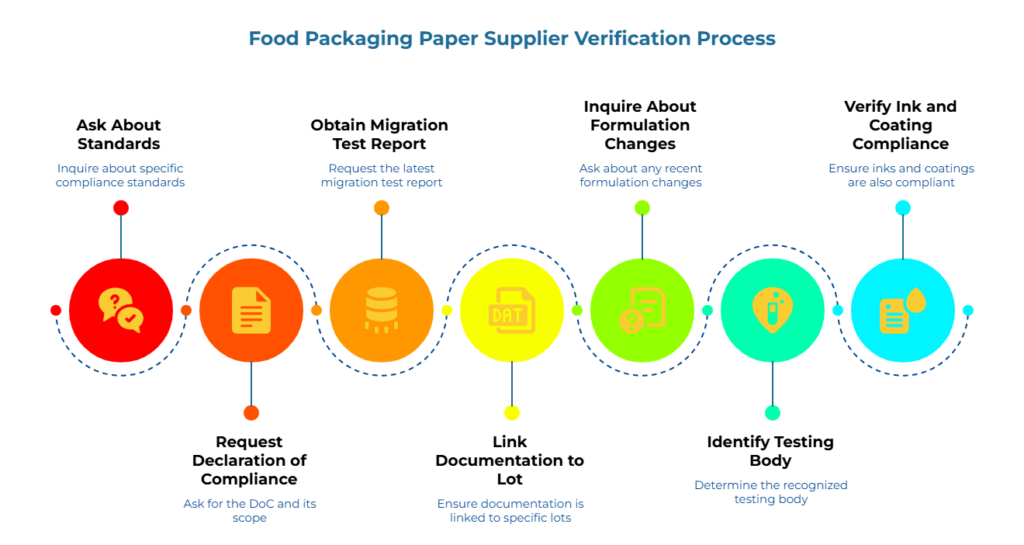
This framework is the centerpiece. A simple set of questions can filter out weak suppliers quickly. As a practical heuristic, this framework can filter out a significant majority of non-compliant offers before resources are committed to price comparisons—complementing broader supplier verification protocols.
1. “Which standard(s) does this material comply with? Name them explicitly.”
Good answer: “This product complies with FDA 21 CFR 176.170 and BfR Recommendation XXXVI.”
Weak answer: “It’s food grade” or “It meets all relevant standards.”
2. “Can you share the Declaration of Compliance and confirm what product grades and use conditions it covers?”
Good answer: Provides a DoC naming specific standards, product codes, and intended food types/temperatures.
Weak answer: “We can send a general certificate” or delayed, vague responses.
3. “Can you share the most recent migration test report, including test conditions?”
Good answer: Provides a dated report from an ISO 17025 accredited lab specifying simulant, time, and temperature parameters.
Weak answer: “Our supplier handles that” or “We don’t have that on file.”
4. “Which lot or COA does this documentation apply to, and how will it be referenced on shipment paperwork?”
Good answer: Clear lot-code linkage between documentation and incoming shipments.
Weak answer: “It applies to all our products” or no lot-level traceability.
5. “Have there been any formulation, coating, or ink changes recently? What was re-tested?”
Good answer: Transparent disclosure with updated documentation.
Weak answer: Silence, or “Nothing has changed” without evidence.
6. “Who performed the testing, and is it a recognized certification body for food-contact materials?”
Good answer: Names a recognized independent testing institute (such as ISEGA) or an ISO 17025accredited laboratory with verifiable credentials—criteria that should be incorporated into your compliance audit checklist for packaging suppliers.
Weak answer: “Internal testing” or inability to name the laboratory.
7. “If the product is printed or laminated, what evidence covers the inks, adhesives, or coatings?”
Good answer: Separate DoC or migration data covering all components in food-contact applications.
Weak answer: “The base paper is compliant” without addressing added layers.
Red Flags: Phrases That Signal Weak Proof
These phrases require immediate follow-up:
| Vague Phrase | Follow-Up Question |
| “Food grade” | “Which standard defines ‘food grade’ for this material?” |
| “FDA approved” | “Which CFR section, and can you share the DoC?” |
| “Certified” | “Certified by whom, for what scope, and when?” |
| “Eco-safe” or “Natural” | “How does this relate to food-contact compliance?” |
| “Same as last time” | “Is the Lot code and formulation identical? Confirm with documentation.” |
These phrases aren’t automatically disqualifying. But the supplier’s response to your follow-up reveals whether you’re dealing with real compliance or marketing filler. Strong suppliers welcome these questions—they differentiate themselves through documentation. Weak suppliers deflect.
For visual guidance on distinguishing authentic certificates from fraudulent ones, see fake FSC, PEFC, ISO certificates vs. real ones: a visual spotter’s guide.
Cheat Sheet: Vague vs. Verified
| Ignore This | Demand This Instead |
| “Food safe” | DoC naming FDA 21 CFR 176.170, BfR XXXVI, or EC 1935/2004 |
| “Food grade” | Specific standard reference with scope statement |
| “FDA approved” | CFR section number plus Declaration of Compliance |
| “Certified” | Certificate from named body with expiration date and scope |
| “Meets all standards” | List of specific standards with supporting documentation |
| “Safe for food contact” | Migration test report with simulant, time, and temperature conditions |
| “Eco-friendly packaging” | Separate food-contact compliance documentation |
| “Same product as before” | Lot-specific COA linking documentation to this shipment |
| “Our supplier confirms it” | Direct documentation from the manufacturer |
| “We’ve never had problems” | Current test reports—not anecdotal history |
Dock-Check Mini Checklist
Before signing for a shipment of food packaging paper, verify:
- DoC attached or on file, naming a specific standard
- Lot code on shipment matches lot code in documentation
- Documentation dated within the last 12–24 months
- Test conditions match your intended use (food type, temperature, hold time)
- Printed/coated materials include component-level compliance evidence
Establishing a Protocol for Continuous Verification
One-off checks aren’t enough. Compliance drifts when suppliers change materials, switch sub-suppliers, or let certifications lapse. A receiving clerk sees “food safe” on a carton; the only way to know is asking for the DoC and the latest migration test tied to that lot. When a health inspector asks for evidence, a neat binder with current certificates reduces anxiety—and risk.
Make documentation a purchase requirement. Include your ask-list in every quote request for food-grade kraft paper, paper bags, or other food-contact materials. Specify that you require a current DoC, migration test report, and lot traceability as conditions of doing business.
For deeper guidance on how compliance breaks down over time, explore why food-safe labels fail and how to verify food contact compliance beyond certificates in the PaperIndex Academy.
Suppliers must provide current, lot-linked compliance documentation as a prerequisite for procurement. Audit-grade documentation ensures that ‘food safe’ claims are substantiated by verifiable data. You can browse food packaging paper suppliers with audit-grade documentation or submit a quote request specifying your documentation requirements upfront.
Frequently Asked Questions
What does “food safe” mean on packaging?
“Food safe” is a general claim, not a standardized certification. Without a named standard and current documents tied to the product and intended use, it should be treated as marketing language rather than proof. Verification requires a Declaration of Compliance naming the specific regulatory framework, a migration test report with relevant conditions, and lot traceability linking documents to the shipment.
Is “FDA approved” the same as “food safe”?
Not necessarily. Useful documentation names a specific regulatory basis (such as 21 CFR § 176.170 for paper and paperboard) and defines the scope in a Declaration of Compliance. Generic wording like “FDA approved” without a CFR reference should trigger follow-up questions about which section applies and whether current documentation exists.
Do buyers always need migration testing?
Migration evidence is commonly used to demonstrate chemical safety under defined conditions, especially when heat, oily foods, coatings, or printing are involved—a topic covered in detail in navigating specific migration limits: a global compliance workflow. The right test conditions—simulant type, time, and temperature—vary by application. A test performed for cold dry goods may not apply to hot greasy foods, so matching test conditions to actual use is essential.
Disclaimer:
This article provides general information about food-contact packaging paper safety claims and documentation for educational purposes. Individual circumstances vary significantly based on factors like market (US vs EU) requirements, intended use conditions (food type, time, temperature), material composition/coatings, and lot-to-lot changes. For personalized guidance tailored to your operation’s compliance and inspection readiness, consult with a qualified professional.
Our Editorial Process:
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team:
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

