📌 Key Takeaways
A 72-hour audit of your supplier vetting process can reveal hidden gaps before defective parent rolls stop your converting line.
- Samples Alone Prove Nothing: A roll that looks good tells you nothing about who made it, whether the factory is real, or if future shipments will match.
- Check the Legal Entity First: Confirm the supplier’s registered company name against a government database before trusting any sample or quote.
- Know Who Actually Makes It: Find out whether you’re buying from a manufacturer, a trader, or a broker—each carries different risks if quality problems arise.
- Connect QA to Qualification Early: Technical requirements like moisture, GSM, and runnability should enter the vetting process before approval, not after a line jam.
- Score Every Supplier Red, Amber, or Green: Use a simple checklist to flag which suppliers were qualified on evidence and which were qualified on assumption.
Documented vetting protects the line; assumptions invite failure.
Procurement managers, QA leads, and operations teams at tissue converting plants will gain a clear audit framework here, preparing them for the step-by-step verification template that follows.
~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~
Industrial converting lines frequently stall due to parent roll defects—such as edge tears or tension failures—that bypass standard visual inspections. These failures often stem from a misalignment between initial supplier qualification and actual delivery integrity.
How did this supplier get approved in the first place?
For cross-border toilet tissue parent roll sourcing, three statements matter:
- A visual sample is not supplier qualification.
- A quote is not supplier qualification.
- A broker introduction is not supplier qualification.
Most toilet tissue converters know this fear. The spreadsheet shows green checkmarks everywhere, but no column confirms whether the legal entity behind the last quote actually exists, whether the factory matches the email signature, or whether the broker who sent the sample has ever set foot in the plant that supposedly made it.
Why Toilet Tissue Converters Should Audit the Vetting Process Before the Next Shipment Crisis
Vetting overseas parent roll suppliers often fails for a predictable reason: the process treats visual samples, responsive emails, and competitive quotes as proof of supplier legitimacy. These elements are not.
A sample that looks and feels right does not confirm who made it. A quote that arrives promptly does not confirm whether the sender is a manufacturer, a trader, or an intermediary who has never touched a parent roll. A business registration number in an email footer does not confirm whether that registration is active, belongs to a paper mill, or connects to the factory that will fulfill the order.
For toilet tissue converters running private-label contracts or regional bathroom tissue operations, weak vetting compounds over time. A supplier that passed informal screening two years ago may have changed ownership, shifted production to a subcontractor, or allowed quality systems to decay. The contract risk surfaces only when a shipment fails incoming inspection—or worse, when defective parent rolls jam the line and halt production continuity.
Rigorous auditing replaces speculative approval with a verification framework rooted in objective data.
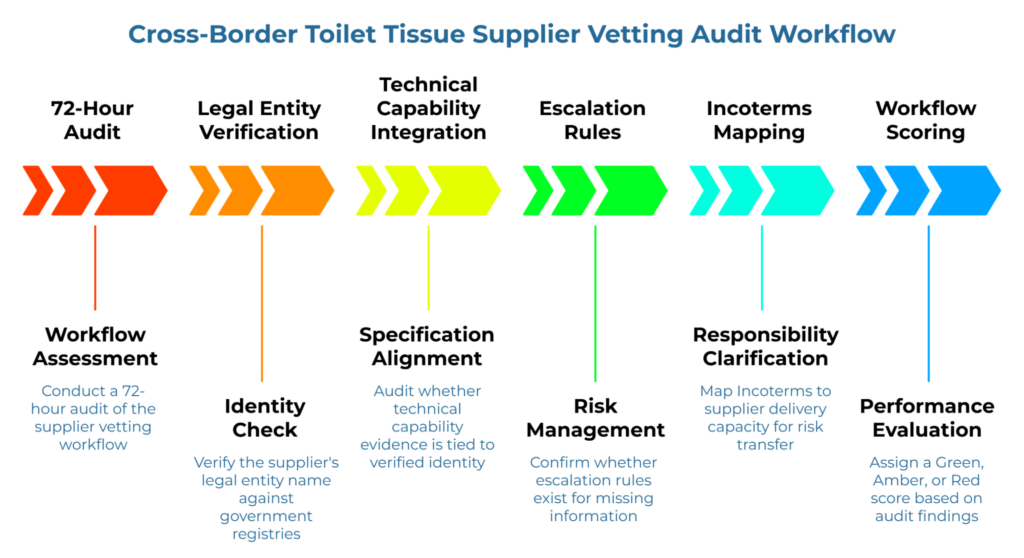
Hours 0–24: Map How a New Overseas Toilet Tissue Supplier Enters Your Process
The first phase documents the current workflow exactly as it operates—not how it should work in theory.
Trace where new toilet tissue parent roll suppliers enter the pipeline. Pull the file for the most recently approved supplier. Where did the lead originate? Common sources include trade shows, online directories, broker introductions, referrals, and inbound inquiries. Each entry point carries different verification requirements. A supplier discovered through a verified directory, such as ‘toilet tissue parent roll suppliers,’ arrives with a different baseline than one introduced by an unknown trading company.
Document every checkpoint, owner, and approval gate in the current process. List every person or department involved in moving a supplier from first contact to approved status. Identify who reviews samples, who checks documents, who signs off on vendor qualification, and who communicates technical requirements. In many tissue converting operations, these responsibilities are distributed informally—supplier onboarding happens by default when no one objects.
For each checkpoint, determine whether it is a documented requirement or an informal habit. As that map takes shape, separate every check into one of two categories: mandatory or informal. Does procurement always request a business license, or only when someone remembers? Does QA review parent roll specifications before qualification, or only after the first shipment arrives? Mapping mandatory versus informal checks reveals where the process relies on individual diligence rather than system design.
Hours 24–48: Identify Verification and Capability Auditing
The second phase stress-tests the evidence chain. The goal is to determine whether the current workflow confirms the supplier’s true identity, rather than assuming it based on surface-level signals.
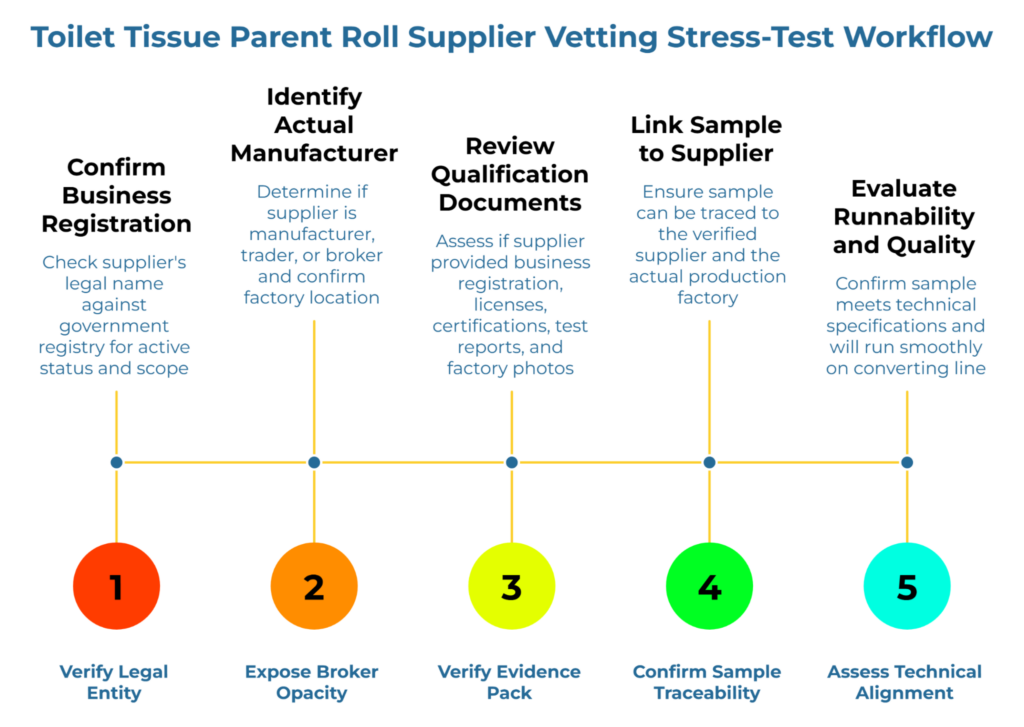
Verify legal entity identity and government registry status. Confirm whether the current vetting process requires verification of the supplier’s legal entity name against a government business registry. For cross-border toilet tissue parent roll sourcing, this step is a regulatory baseline. Government registry cross-referencing confirms that the company exists, that its registration is active, and that its stated business scope includes paper manufacturing or trading.
A commercial identity and a legal identity are not always the same. A brand name, email signature, or sample label can feel reassuring while still leaving the underlying entity unclear. That gap becomes harder to fix later, especially if claims, complaints, or technical failures emerge after shipment.
If the workflow skips this compliance check, the process has a structural gap. Established due diligence frameworks treat legal entity verification as a baseline requirement for responsible sourcing.
Expose broker or trader opacity and confirm who actually makes the toilet tissue parent rolls. Determine whether the current workflow distinguishes between manufacturers, traders, and brokers. A manufacturer operates a mill and controls production. A trader buys and resells without manufacturing capability. A broker facilitates introductions and may never handle products. Each role is legitimate, but buyer risk differs dramatically.
When converters believe they are working with a manufacturer but the actual supply comes through an undisclosed intermediary, accountability for quality problems becomes unclear. Technical capability evidence cannot be verified. Specification alignment becomes harder to enforce. The buyer loses visibility into who is responsible when parent rolls arrive with moisture out of spec, GSM inconsistency, or bulk that won’t run cleanly.
Pull the file for a recently approved supplier and identify the actual factory. If the factory name, location, and contact are documented, the workflow has some transparency. If the file contains only a trading company name and the factory remains unknown, the process is exposed to downstream cross-border risk.
Verify the evidence pack required for qualification. Review the evidence pack for a recently approved supplier. Strong toilet tissue parent roll supplier vetting typically requires business registration documents, export licenses where applicable, quality management certifications such as ISO 9001, product test reports, and factory photographs or audit records. If the current workflow accepts verbal assurances or sample approval alone, the process lacks the documentation needed to hold a supplier accountable.
A sample proves nothing if it cannot be traced to the verified supplier. Determine whether the current workflow confirms sample-to-supplier traceability—that the sample came from the factory that will fulfill production orders. Without this link, a converter may qualify a supplier based on a sample sourced from a different mill entirely.
Visual inspection of a toilet tissue parent roll can confirm surface quality, winding, and basic appearance. It cannot confirm GSM consistency across production runs, moisture tolerance under shipping conditions, bulk stability over time, or runnability on the converting line. If the current workflow treats visual approval as the final qualification step, technical alignment gaps will surface later—often during production.
Hours 48–72: Check Whether Technical Alignment Is Embedded in Vetting
The third phase audits whether the current vetting workflow connects supplier qualification to technical capability before approval—not after trouble starts.
Audit whether technical capability evidence and escalation rules are tied to the verified supplier identity, then score the workflow. Do not turn this step into a full technical-specification review. Instead, ask a workflow question: where do toilet tissue parent roll requirements enter the qualification sequence?
In a strong workflow, toilet tissue parent roll specifications—physical properties, moisture levels, and mechanical performance specifications—are communicated before qualification and confirmed through quality documents or testing. If QA requirements appear only after the first production problem, the workflow treats qualification and technical review as separate processes. This separation creates risk.
Determine whether the workflow transfers supplier evidence from procurement to QA in a structured way. When qualification happens in one department and technical review happens in another without shared documentation, gaps emerge. A strong process ensures that the legal entity name, factory identity, sample traceability, and specification alignment travel together from vetting through ongoing supply.
The audit should also confirm whether escalation rules exist. If a field is missing—legal entity unconfirmed, manufacturer unknown, capability evidence absent—does the workflow define what happens next? Or does the supplier advance to the approved supplier list anyway?
For cross-border trade, understanding how responsibilities shift between supplier, freight forwarder, and buyer is essential. The ICC Incoterms® 2020 rules clarify risk transfer points and must be mapped to the supplier’s declared delivery capacity to ensure technical specifications are met at the point of transfer.
Workflow Scoring Matrix
At the end of the 72-hour audit, assign a score to the current cross-border toilet tissue supplier vetting process:
Green: The workflow confirms legal entity identity, checks government registry status, distinguishes manufacturer from trader from broker, requires documented capability evidence, connects sample traceability to the verified supplier, and triggers QA review before qualification. Escalation rules are defined.
Amber: The workflow includes some checks but relies on informal execution, lacks documentation for key steps, or does not consistently distinguish manufacturers from intermediaries. Some suppliers on the approved list may not have been fully verified.
Red: The workflow treats visual sample approval and responsive communication as qualification. Legal identity, registry status, and broker transparency are not systematically confirmed. Technical alignment enters the process only after transit failure or line stoppage.
Quick-Audit Template: Cross-Border Toilet Tissue Supplier Vetting
Use this checklist to score each supplier in the current pipeline:
- Supplier source documented
- Named legal entity confirmed
- Government registry status checked
- Actual manufacturer identified
- Broker/trader involvement disclosed
- Primary contact traceable to verified entity
- Sample traceable to verified supplier
- Quality/technical evidence requested
- Factory capability evidence requested
- Escalation rule defined if any field is missing
- Final assessment category
A supplier missing essential identity documentation, or traceability, generally warrants immediate review. This template can support ongoing vendor qualification and supply continuity monitoring.
The Five Fixes That Most Quickly Improve a Weak Cross-Border Vetting Process
For toilet tissue converters whose audit reveals amber or red scores, five core upgrades are estimated to deliver measurable operational improvement:
- Separate supplier discovery from supplier qualification. Finding a potential toilet tissue parent roll supplier is not the same as proving that supplier is trustworthy and capable. Require a qualification gate between discovery and approval.
- Require legal entity confirmation before sample trust. Confirm that the company name, registration number, and business scope match before evaluating a sample. A sample from an unverified entity proves nothing about production continuity.
- Make government registry cross-referencing mandatory. Check the supplier’s registration status against an official government business registry. Document the result. Skip this step, and the process cannot confirm the supplier legally exists.
- Document who the actual manufacturer is. If a broker or trader is involved, require disclosure and confirm the identity of the factory. Accountability for quality depends on knowing who made the toilet tissue parent rolls.
- Connect technical review to supplier qualification before approval. Ensure that QA requirements — GSM, moisture tolerance, bulk, runnability—are communicated and confirmed as part of vetting, not treated as a separate downstream process. A practical framework for normalizing specifications can support this handoff, and embedding ISO 287 testing standards in RFQ language strengthens specification alignment from the start.
A documented, multi-layer cross-border toilet tissue supplier vetting process does not guarantee perfect suppliers. It ensures that qualification decisions rest on verified evidence—safeguarding conversion continuity while providing procurement and QA teams a defensible, unified operational baseline.
If the current workflow scores lower than expected, review toilet tissue raw materials suppliers, browse more guidance in the PaperIndex Academy, or find suppliers using the same qualification logic from the start.
Frequently Asked Questions
What should be checked before approving a new overseas toilet tissue parent roll supplier?
Before approval, confirm the supplier’s legal entity name against a government registry, verify whether the supplier is the actual manufacturer or an intermediary, request capability evidence and quality documents, and ensure sample traceability back to the verified factory.
Why are visual samples not enough for toilet tissue supplier qualification?
Visual inspection confirms surface quality and winding but cannot verify GSM consistency, moisture tolerance, bulk stability, or runnability across production runs. A sample may also originate from a different source than the factory that will fulfill orders.
How do you verify whether a cross-border supplier is the actual manufacturer?
Request documentation that names the factory, its location, and its direct contact. Cross-reference this against business registry records. If the supplier cannot disclose the manufacturing source, broker or trader opacity may be present.
When should government registry checks happen in supplier vetting?
Registry checks should occur before sample evaluation and before any supplier advances to the approved supplier list. Confirming legal entity status early prevents qualification based on unverified claims.
How can procurement and QA share the same supplier-vetting baseline?
Ensure that the evidence pack—legal entity confirmation, factory identity, sample traceability, and technical capability documents—travels from procurement through QA review. Define escalation rules so both teams know what happens when documentation is incomplete.
Disclaimer
This content is for informational purposes only and does not constitute legal, procurement, or compliance advice. Supplier vetting frameworks, registry verification approaches, and audit workflows are illustrative; actual requirements vary by jurisdiction, product type, and organizational context. Outcomes depend on implementation quality and supplier cooperation. Consult qualified legal, trade compliance, and supply chain professionals before modifying vetting processes or making sourcing decisions.
Our Editorial Process:
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team:
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

