📌 Key Takeaways
“Pharma-grade” sounds precise but leaves the board properties that determine code readability completely undefined and that ambiguity causes scanner failures.
- Generic Labels Hide Real Gaps: “Pharma-grade” is commercial shorthand, not a technical specification—suppliers interpret it differently, creating hidden mismatches.
- Scanner Failures Start in Quote Language: Most readability problems trace back to vague specifications, not printing mistakes or faulty equipment.
- Normalize Before You Compare: Define board behavior, surface properties, and print tolerances so every supplier quotes the exact same technical job.
- Specify What the Board Must Do: Shift from asking “what category is this?” to “what must this accomplish?”—then name the test methods that prove it.
- Quote Requests Are Quality Checkpoints: If readability requirements are missing from the original request, compliance risk is already built in before production starts.
Specification-true control eliminates the guesswork that turns normal variability into systematic failure.
Pharmaceutical packaging buyers and procurement managers will gain a clear framework for preventing readability audit failures, preparing them for the detailed specification guidance that follows.
~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~
“If a board is quoted as pharma-grade, it should be capable of supporting readable 2D codes.”
This assumption makes intuitive sense. The term “pharma-grade” sounds like a rigorous standard—something with regulatory weight behind it. When a supplier confirms that their board meets pharmaceutical packaging requirements, it seems reasonable to expect that board will support the security features modern serialization systems demand.
And yet, folding carton shipments quoted as pharma-grade routinely fail readability audits. The 2D DataMatrix codes look acceptable during visual inspection. The certificate of analysis confirms specification compliance. But when those folding cartons reach the verification station, high-speed scanners struggle to decode them consistently enough to keep the line running.
The problem is not careless suppliers or faulty equipment. The problem is that “pharma-grade” is not a control variable. It is commercial shorthand, language that sounds authoritative while leaving the physical and print-performance variables that actually determine scanner readability completely undefined.
Barrier Property Normalization offers a different approach: the precise documentation of board grades, barrier properties, and printing tolerances required to support mandatory pharmaceutical security features. Think of it like pouring the exact right concrete foundation before installing a high-tech security system. Without that foundation specified correctly, even the most sophisticated system will fail.
Why “Pharma-Grade” Is A Commercial Shorthand, Not A Production-Ready Specification
The term “pharma-grade” carries an implied promise of quality and regulatory alignment. In practice, however, no regulatory body defines it as a controlled specification for folding carton board.
The FDA’s Drug Supply Chain Security Act (DSCSA) and the European Commission’s Falsified Medicines Directive (FMD) establish the legal framework for interoperable, electronic tracing of products at the package level. While these regulations mandate the presence and quality of the ‘Unique Identifier’ (UI) and ‘Data Matrix’—often pointing to technical standards like ISO/IEC 16022 and ISO/IEC 15415 for print quality—they remain technologically neutral regarding specific substrate material properties. It is the responsibility of the manufacturer to ensure the chosen board properties (such as brightness, opacity, and gloss) are compatible with these ISO-standardized readability requirements.
This creates a translation gap. When a quote request asks for “pharma-grade board,” different suppliers interpret that phrase through different lenses. One focuses on virgin fiber content. Another prioritizes surface smoothness. A third emphasizes moisture barrier coatings. Each interpretation may be legitimate for certain applications, but none guarantees that the resulting board will support consistent 2D code readability at production line speeds.
The variables that determine whether a DataMatrix code scans reliably—porosity levels, ink absorption characteristics, surface energy, caliper consistency—remain unaddressed. Suppliers fill this specification vacuum with assumptions. Those assumptions become invisible until scanner failures reveal them.
That is why vague requirements destroy quote comparability. When suppliers interpret the same request differently, the resulting quotes cannot be evaluated on an equivalent basis—a problem that demands systematic quote normalization before any commercial evaluation begins. Significant variance in quoted values—often exceeding the ± 5\% typical of virgin fiber market fluctuations—signals a fundamental specification mismatch. When one supplier assumes a high-performance aqueous coating for ink-holdout while another quotes a standard uncoated SBS, the resulting delta reflects a difference in the technical job being performed, not just a competitive margin. The spread may be telling the buyer that different suppliers are not quoting the same technical job at all.
The Anatomy Of A Failed Scan
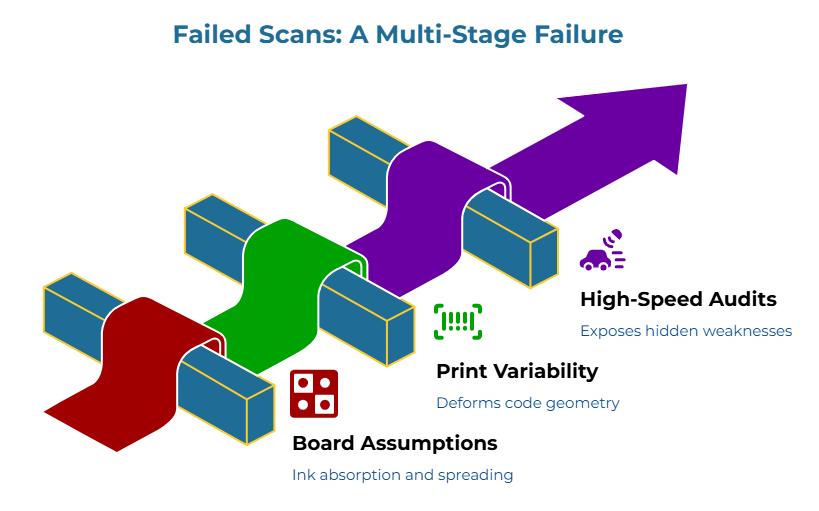
Understanding why generic specifications fail requires tracing the physical chain from board selection to scanner rejection. The failure mechanism operates through three distinct stages, each amplifying the consequences of upstream ambiguity:
Stage 1: Board assumptions alter how ink sits, spreads, or absorbs. When suppliers select boards based on generic “pharma-grade” language, they make implicit decisions about substrate characteristics. A board with higher porosity absorbs ink differently than one with tighter fiber density. A board that absorbs too aggressively can soften edges. A surface that handles ink differently than expected can widen modules or reduce crispness. Surface treatments alter how ink spreads and sets. These are not defects—they are simply properties that differ from what the printing process requires for fine-resolution codes.
Stage 2: Print variability deforms the code or micro-text. DataMatrix codes depend on precise geometric relationships. The GS1 DataMatrix guidelines specify module dimensions, quiet zones, and contrast requirements that demand tight control over print output. When ink absorbs unevenly or spreads inconsistently, individual modules in the matrix expand or blur. The printed code may look acceptable to the eye while its underlying geometry has shifted beyond scanner tolerance. This is where many teams lose the argument internally. The print room sees a code. Quality sees intermittent failures. Procurement sees competing explanations. The code becomes a troubleshooting problem when it was really a specification problem from the start.
Stage 3: High-speed readability audits expose the hidden weakness. Production-line scanners verify codes in milliseconds. Marginal codes that decode correctly at slow speeds or optimal angles fail when those tolerances tighten under real production conditions. The readability audit reveals what human inspection cannot see: the codes meet minimum thresholds only under ideal conditions that production environments do not provide.
What Readability Audits Are Really Revealing
A failed readability audit typically triggers an investigation focused on the print house, ink supplier, scanner calibration, or artwork files. These investigations often find nothing definitively wrong because the root cause lies outside their scope.
The audit is revealing a specification failure, not an execution failure.
When the quote request used generic terminology, every downstream participant made reasonable choices based on incomplete information. The board supplier selected a substrate that met their understanding of the requirements. The converter calibrated to the board they received. The verification system assessed the codes as printed. Each step was executed competently within its local constraints. The failure emerged from the gap between what was specified and what was actually needed.
For broad regulatory context on why package-level traceability and safety features matter, the official references are the FDA’s DSCSA overview and the European Commission’s falsified medicines framework. For symbol implementation and reading context, the GS1 DataMatrix User Guide is the core technical reference.
This shift in perspective ensures that verification success is engineered into the procurement phase, rather than left to chance at the final inspection point.
Barrier Property Normalization: The Missing Discipline Behind Reliable Scans
Barrier Property Normalization replaces inference with control.
Instead of requesting “pharma-grade board,” the specification defines the barrier characteristics, surface properties, and print-performance parameters that the application actually requires. This approach inverts the typical quote process. Rather than describing what the board should be called, it describes what the board must do. Suppliers quote against functional requirements they can test and verify. Comparisons between suppliers become meaningful because everyone interprets the same specification language the same way.
For serialization-critical applications, normalization means specifying ink holdout characteristics that support the module dimensions and contrast ratios the verification system requires. It means defining porosity ranges that prevent ink spread beyond acceptable tolerances. It means establishing surface energy parameters that ensure consistent ink transfer across the sheet and across production runs.
The discipline is straightforward in concept. It simply requires shifting from procurement language to engineering language—from “what category does this board belong to” to “what must this board accomplish.” Implementing a specification-true normalization matrix before evaluating any supplier quotes transforms the entire downstream process—a methodology formalized in the proof ladder for folding carton suppliers.
The Specification Fields Suppliers Should Never Have To Guess
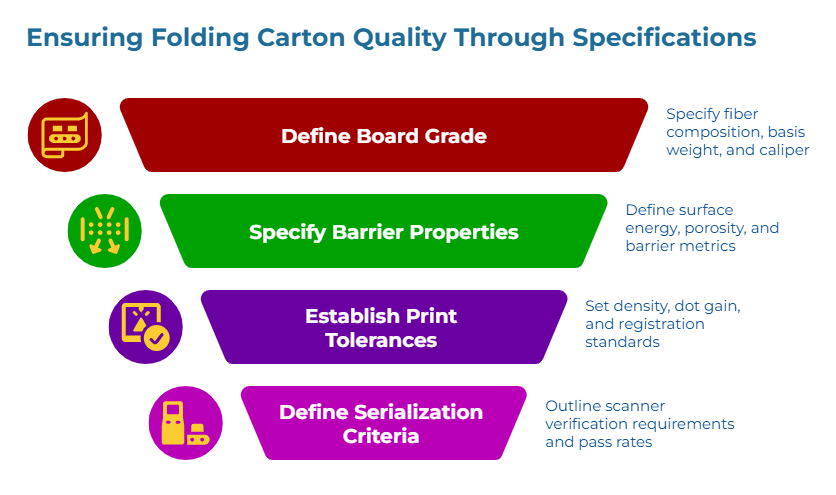
Moving from generic labels to specification-true quotes requires defining the parameters that govern print and readability performance. The following fields represent the minimum specification set for serialization-critical folding cartons.
Board Grade And Substrate Behavior
Specifications should define fiber composition requirements, basis weight with tolerances, caliper ranges, and permitted caliper variation across the sheet. These parameters affect print pressure, ink transfer, and dimensional stability through converting operations.
A specification stating ‘SBS board, 280–300 g/m²’ provides more actionable guidance than ‘pharma-grade board.’ A specification adding ‘caliper 350–380 µm, maximum cross-direction caliper variation ±5 µm’ eliminates most substrate-related print variability before it starts—a principle explored further in board grade tolerances explained: securing folding carton specifications across suppliers.
Barrier Properties And Surface Behavior
Surface characteristics directly influence how ink behaves during and after printing. Specifications should define surface energy ranges, porosity or air permeability values, and surface treatment requirements as applicable to the printing method and end-use environment.
For applications requiring moisture or product barriers, specifications should include Cobb values, water vapor transmission rates, or other barrier metrics appropriate to the distribution conditions the folding carton will encounter. Related guidance on mapping folding carton barrier requirements to your budget can help align these parameters with actual product needs.
Print Tolerance Baselines And Test Methods
Specifications should establish acceptable ranges for print density, dot gain, registration tolerances, and color variation. Critically, the specification must name the test methods used to verify these parameters. Without a defined measurement standard (such as ISO 5-3 or 5-4), a density target remains an unenforceable subjective preference.
For DataMatrix codes printed on folding cartons, specifications should reference ISO/IEC 15415 for label-based applications or ISO/IEC 29158 (AIM DPM) if the code is applied via direct marking that might involve substrate-specific reflectance challenges. Establishing a minimum ‘Grade C’ or higher under these standards ensures that the interaction between the board’s surface energy and the ink’s spread remains within a decodable range for high-speed line scanners.
Teams establishing what documentation to require at the quotation stage can reference what proof to request at RFQ stage for folding cartons for a structured evidence framework.
This connects board and print specifications directly to the verification requirements that determine pass or fail.
Serialization And Readability Acceptance Criteria
The specification should define scanner verification requirements: minimum grade thresholds, scan angle tolerances, lighting conditions, and pass rates at production speeds. These criteria flow backward into the print and board specifications, ensuring alignment between what is ordered and what is needed.
Including readability acceptance criteria in the board specification makes the relationship explicit. Suppliers understand that “pharma-grade” is not an acceptable substitute when the actual verification requirements are stated in measurable terms. For teams trying to make quote logic more consistent, how to normalize your first folding carton quote: a beginner’s guide offers a useful framework. PaperIndex Academy is helpful here because it frames specification normalization as an educational discipline, not a sales pitch.
Myth Vs. Reality: The Four Assumptions That Keep Failing On The Line
Myth 1: “Pharma-grade is enough.” Reality: “Pharma-grade” is undefined commercial language that different suppliers interpret differently. It signals intent, not full operational control. Consistent results require consistent specifications with named parameters and test methods.
Myth 2: “If one supplier can print it, any supplier can.” Reality: Readability depends on the interaction between specific board properties and specific print processes. A board that performs well with one converter’s equipment configuration may fail with another’s. Shared output claims do not prove shared substrate assumptions.
Myth 3: “Scanner failure is a printing-room issue only.” Reality: Most scanner failures trace to board properties that printing cannot correct. The scanner is exposing a mismatch that often began upstream, in the way the board and surface were specified. The fix belongs in the specification, not in press-room adjustments after the fact.
Myth 4: “Compliance verification starts after the quote is awarded.” Reality: The quote request is the first compliance checkpoint. If readability expectations are absent from the quotation logic, the compliance risk is already embedded before the first production run. Specifications that cannot support verification requirements will fail regardless of how well suppliers execute against them.
What To Change Before The Next Quote Round
The shift from generic to specification-true quoting does not require overhauling procurement systems. It requires adding precision at specific points in existing processes. The next specification cycle should follow four steps in sequence:
First, replace “pharma-grade” with specification language that defines board behavior, barrier and surface behavior, print tolerance baselines, and readability expectations. Start by documenting the readability requirements the serialization system actually enforces. Work with quality and validation teams to establish minimum acceptable grades, pass rates, and test conditions. These become the anchor specifications that all other parameters must support.
Second, normalize those requirements across all suppliers so the quoted job is technically the same job—following the approach outlined in the folding carton specification alignment checklist. Translate readability requirements into board and print specifications. Identify the substrate properties that enable the print performance required. Resources available through the PaperIndex Academy and guides on quote normalization can support this translation process.
Third, establish the verification protocols and acceptance criteria that will be used to evaluate readability performance. Build a specification template covering board composition, basis weight and caliper ranges with tolerances, surface treatment requirements, barrier property values, print specification baselines, and readability acceptance criteria. Name the test methods for each verifiable parameter.
Fourth, update quote evaluation to compare submissions against normalized specifications rather than against each other. Resolve interpretation gaps before evaluating anything else. The specification-true quoting framework provides additional structure for this comparison process.
That sequence matters. First define the job. Then compare suppliers. Then validate execution. For organizations managing pharmaceutical supply chain compliance more broadly, related guidance on protecting your pharmaceutical supply chain addresses the governance context surrounding specification discipline.
Move From Generic Labels To Specification-True Control
Readability failures in pharmaceutical packaging rarely originate at the verification station. They originate in the specification gap—the space where “pharma-grade” substitutes for the detailed, measurable parameters that actually govern scanner performance.
The scanner that rejects folding cartons is not malfunctioning. It is identifying codes that fail to meet verification thresholds because the board could not support the print precision those codes required. The board could not support that precision because the quote request never specified it.
Closing that gap means treating ‘pharma-grade’ as a starting point for conversation rather than a finished specification—an approach detailed in from guesswork to governance: a comprehensive folding carton supplier verification methodology. It means defining board behavior, barrier properties, print tolerances, and readability criteria in language that suppliers can verify, test, and guarantee. It means treating the quote request as the first quality checkpoint rather than administrative paperwork.
Replace shorthand with Specification-True Folding Carton Blueprints. Replace interpretation with Barrier Property Normalization. Replace late-stage surprises with earlier control.
Normalized requirements reduce downstream QA failure risk by ensuring everyone—buyer, supplier, converter, verification system—operates from the same factual baseline. Specification-true control does not eliminate all variability. It eliminates the guesswork that turns normal variability into systematic failure.
Readable 2D codes depend on disciplined upstream definition. The exact mix of board behavior, barrier behavior, surface behavior, and print tolerance baselines will vary by application, process, and market. The need for explicit control will not.
Readability reliability begins in the quote language, not at the audit station.
Disclaimer
This article is for informational purposes only and should not be treated as legal, regulatory, validation, or quality-release advice. Packaging, serialization, and readability requirements vary by product, substrate, printing process, destination market, and applicable law. Final specification and release decisions should be reviewed by qualified packaging, quality, regulatory, and supply-chain professionals.
Our Editorial Process:
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team:
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

