📌 Key Takeaways
A 72-hour audit of your pharmaceutical folding carton specifications stops suppliers from making decisions that should be yours.
- Old Files Fail Quietly: Specifications that look fine can hide vague board grades, missing barrier requirements, and print language too loose for modern drug packaging rules.
- Gather Everything First: Pull every document your team actually uses—RFQ templates, legacy sheets, supplier data sheets, even email threads that clarify requirements.
- Check Material Fields: Board grade, barrier properties, and coatings must be stated clearly enough that two suppliers reading the same file reach the same conclusions.
- Print Specifications Need Precision: Generic “barcode ready” language won’t meet today’s serialization rules—your specifications should support 2D matrix readability with explicit tolerances.
- Consolidate Into One Baseline: A single, buyer-owned specification document removes supplier guesswork and makes quotes actually comparable.
Vague specifications transfer control to suppliers. Clear specifications keep control with you.
Packaging, compliance, and quality teams preparing for supplier outreach will find a practical three-phase audit framework here, setting up the detailed checklist and next-step resources that follow.
~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~
A practical starting framework for packaging, compliance, and quality teams to assess whether their current folding carton specification files are complete enough for modern pharmaceutical requirements before supplier outreach begins.
A rigorous audit systematically addresses file collection, board-grade verification, barrier-property analysis, print-tolerance documentation, and baseline consolidation—ultimately enabling buyers to source folding cartons with confidence.
Pharmaceutical folding carton specifications can be audited in phases:
- Gather all current folding carton specification and RFQ files into one working set.
- Check for missing board-grade fields.
- Check for missing barrier-property fields.
- Check whether print tolerance requirements for 2D matrices are documented.
- Consolidate every gap into one updated specification-true baseline.
Why a 72-Hour Audit Matters Before You Send Another Supplier Request
Legacy specification files have a way of looking adequate. They sit in shared drives, get attached to RFQs, and produce quotes that arrive on schedule. The problem surfaces later—when two suppliers interpret the same document differently, and the quotes that seemed comparable turn out to reflect entirely different assumptions about board grades, barrier properties, or print capabilities.
Old files rarely fail in dramatic ways. They fail quietly. A board grade is implied instead of named. A coating is assumed instead of specified. When it comes time to compare quotes from folding carton suppliers, those unstated assumptions become the root cause of incomparable responses. A print note says “barcode-ready” even though current pharmaceutical packaging specifications need tighter language around 2D matrix readability and other print-critical controls.
Outdated pharmaceutical folding carton specifications create a specific kind of risk. They do not announce themselves. Instead, they quietly transfer decision-making authority from the buyer’s team to supplier interpretation. One supplier assumes a particular caliper. Another assumes a different coating. Neither is wrong based on what the file actually stated. Both are quoting different folding cartons.
A focused 72-hour audit addresses this before the next supplier round begins. The goal is not to rewrite every document from scratch. It is to surface the gaps that matter most—the unstated board grades, the assumed barrier properties, the vague print language—and consolidate them into a single, defensible baseline.
This specification risk is especially relevant in pharmaceutical and healthcare packaging, where packaging documentation may need to support downstream security and traceability requirements. In the United States, the Drug Supply Chain Security Act (DSCSA) establishes requirements for product identifiers on prescription drug packages, defining the established requirements for the electronic identification and tracing of certain prescription drugs at the package level. In the European Union, Commission Delegated Regulation 2016/161 mandates unique identifiers and anti-tampering devices as part of the Falsified Medicines Directive framework, setting out the safety features, how authenticity should be verified, and by whom.
The operative requirement is clear: if the file set is not explicit, the supplier response will not be fully comparable.
Inventory and Centralization of Active Files
The first phase is collection. Pull together every document your organization currently treats as “the specification” for pharmaceutical folding cartons—not just the files that should be authoritative, but the ones that actually get used.
This includes active RFQ templates currently sent to suppliers, internal board-grade specifications maintained by packaging engineering, print files and artwork guidelines referenced during production, legacy packaging-specification sheets still circulating in operations, supplier-originated technical data sheets that have been adopted as working standards, and version-controlled internal notes or email threads that clarify requirements not captured elsewhere.
The purpose is visibility. The point is not elegance. Many organizations discover during this phase that their official house specification differs from what actually gets attached to supplier requests. Others find that critical requirements live in email threads rather than formal documents. Still others realize that a supplier’s data sheet has become the de facto baseline—meaning the supplier, not the buyer, controls the specification. This pattern often emerges when teams have not systematically evaluated folding carton suppliers against buyer-owned criteria.
Place everything in one working folder. This becomes the audit set for the next 48 hours. Do not filter or organize yet. The goal is to see the true documentation landscape before evaluating it.
During this first pass, sort each file by three questions: Is it active? Who uses it? What decision does it influence? That turns a pile of documents into an auditable working set and establishes the threshold for moving into field-level review.
Material Integrity and Barrier Verification
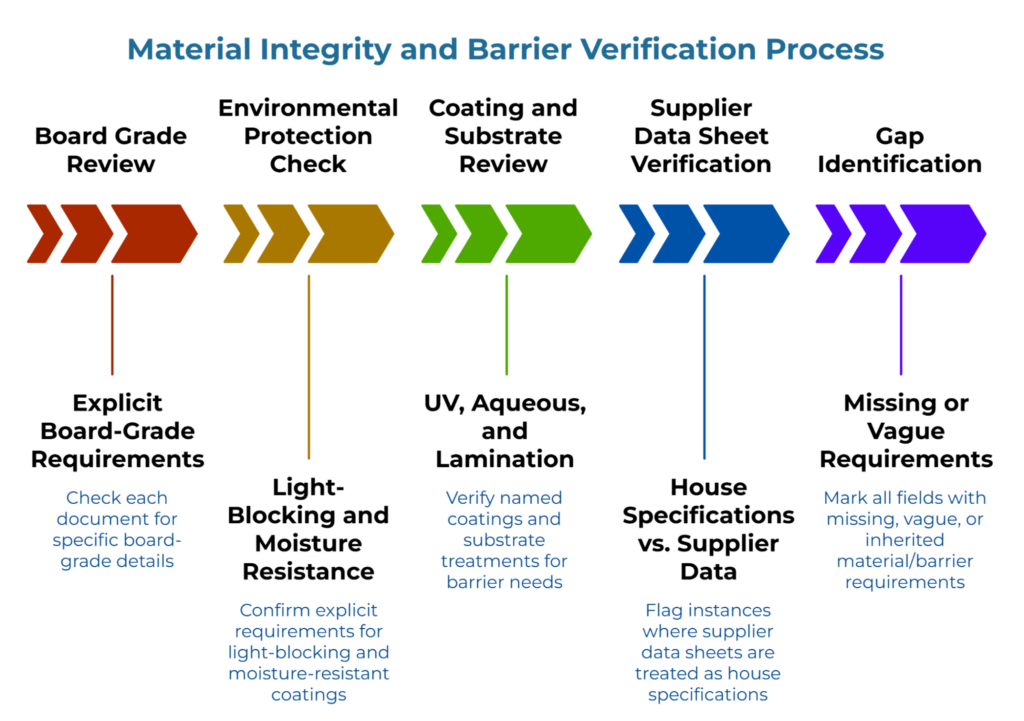
With the audit set assembled, the second phase focuses on material and barrier specifications—the fields that create the most supplier variance when left unstated. A file is not ready because it looks complete. It is ready only when the material baseline is explicit enough to produce consistent interpretation.
Start with board grades. Review each document for explicit board-grade requirements. If files reference generic terms like “SBS” or “folding boxboard” without specifying caliper, basis weight, or grade family, folding carton suppliers will interpret them differently. Those interpretations become quote variance and, eventually, production inconsistency—a pattern analyzed in the lowest bidder myth: why cheap folding cartons cause production downtime. For guidance on how board-grade ambiguity affects supplier quotes, see board grade tolerances explained: securing folding carton specifications across suppliers.
Then check necessary environmental protections. Confirm whether light-blocking requirements or specialized moisture-resistant coatings are stated explicitly. While primary packaging (like blisters or bottles) typically handles oxygen and moisture vapor barriers, pharmaceutical secondary folding cartons often require specific light performance and physical durability that varies by product type and storage conditions. If these secondary-specific requirements remain unstated, suppliers may quote materials that meet basic physical needs but fail to protect the primary container adequately throughout the supply chain. Broad protective language can hide materially different interpretations.
Next, review coatings and substrate treatments. Confirm whether UV coatings, aqueous coatings, or laminations are named where applicable. For pharmaceutical applications where barriers must protect primary containers, alignment with the principles in mapping folding carton barrier requirements to your budget: the hidden cost of over-packaging can help prevent over-specification. Unstated coating expectations frequently surface as post-production disputes. Surface treatment affects both protection and print behavior.
Finally, check for supplier-provided data sheets being used as house specifications. Flag any instance where the team treats a supplier’s technical data sheet as the working specification. This arrangement transfers baseline control to an external party. When that supplier updates their sheet—or when you switch suppliers—your specification changes without internal approval. The risks of this practice are explored in the truth decay of data sheets: why relying on supplier specifications ruins folding carton specifications.
Mark every field where material or barrier requirements are missing, vague, or inherited from supplier documents. These gaps form the correction list for the final phase. This phase is less about theoretical completeness than about removing the silence that lets suppliers fill in the blanks.
Standardization of Serialization and Print Controls
The third phase addresses print-related specifications. The question is not whether a folding carton can carry a code. The question is whether the documentation is precise enough to support reliable code reproduction.
Regulatory frameworks, such as the FDA’s DSCSA, now mandate specific product identifiers for pharmaceutical packaging. These regulatory requirements translate into specific demands on folding carton specifications.
Check 2D matrix readability requirements. Confirm whether current specifications state requirements that support 2D DataMatrix readability—or whether they use generic “barcode” language that leaves critical parameters undefined. The GS1 General Specifications, operating in strict conjunction with the ISO/IEC 15415 standard, defines the technical parameters for 2D symbol quality, quiet zones, and print contrast grading. Folding carton specifications should be explicit enough for suppliers to meet these standards without guessing.
Review print tolerances. Check whether print-critical tolerances are documented clearly enough for supplier interpretation. Vague language such as “high-quality print” invites variance. Explicit tolerances—registration accuracy, minimum line weights, contrast requirements—eliminate ambiguity and create enforceable baselines. Terms such as “barcode capable” do not define what the converter is actually expected to hold.
Confirm verification expectations. Check whether test methods or verification expectations are named where relevant. If downstream processes require specific scan verification grades or readability thresholds, upstream specifications should state those requirements.
Specific tolerances, verification methods, and symbol-quality controls can vary by product, market, equipment, and internal quality system. The practical test is whether current files name those expectations clearly enough for the next supplier conversation. This phase connects documentation quality to downstream compliance readiness. Gaps here create more than quote inconsistency—they create audit exposure.
Turn The Gaps Into A Specification-True Folding Carton Blueprint
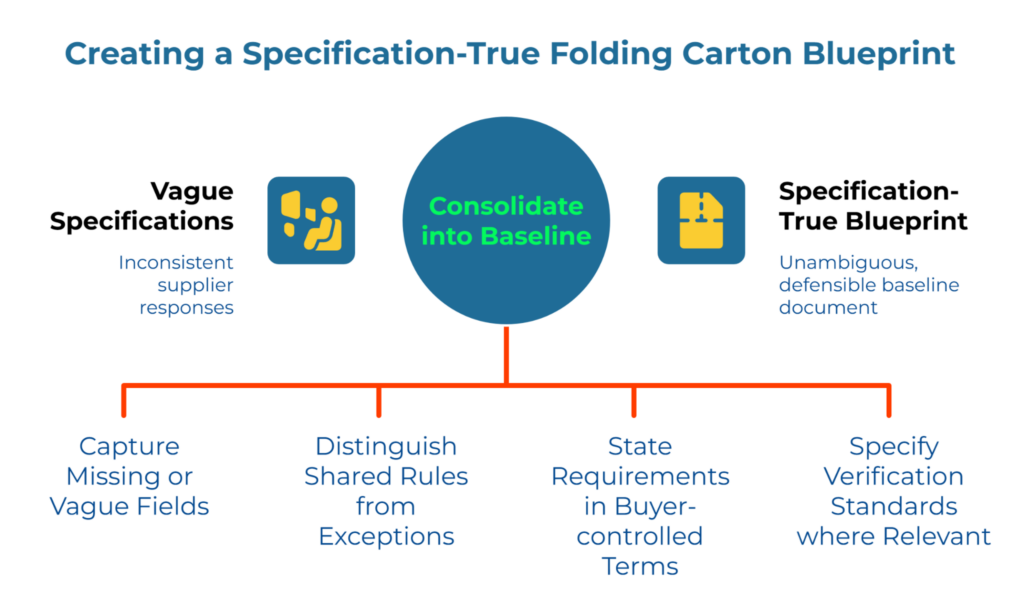
By hour 72, the audit has produced a list of gaps across material, barrier, and print-readiness fields. The final step is consolidation.
Create a single working document—a unified technical standard—that captures every missing or vague field identified during the audit. For guidance on structuring this baseline, refer to the baseline packaging parameter checklist: structuring your folding carton specification requirements. This becomes the buyer-owned blueprint for future supplier interactions.
This baseline must capture approved board grades, barrier properties, coatings, and print-readiness requirements, clearly distinguishing between shared rules and SKU-specific exceptions. Remove duplicate or conflicting language and retire any supplier data sheet that has been standing in for the actual house specification.
Consolidation Principles:
Separate shared requirements from SKU-specific exceptions. A single baseline document can serve multiple products if the distinction between universal requirements and product-specific variations is clearly maintained. State requirements in buyer-controlled language rather than inherited supplier terminology. Include the test methods or verification standards that matter to downstream processes where practical and relevant.
A specification-true folding carton blueprint does not need to be complicated. It needs to be unambiguous. That is what turns scattered legacy files into a working baseline that is easier to compare, easier to update, and easier to defend.
Maintain a disciplined workflow: initial collection, field-level verification, and final consolidation. That sequence restores control without expanding the audit into a full redesign exercise.
The goal is not a perfect document. It is a defensible baseline that eliminates the most dangerous ambiguities before the next supplier conversation. This baseline enables quote normalization—the process of ensuring that supplier responses reflect the same underlying assumptions. Whether working with domestic converters or international folding carton manufacturers, the same principle applies. For guidance on applying a specification-true baseline to quote evaluation, see how to normalize your first folding carton quote: a beginner’s guide.
Understanding why vague specifications create quote variance in the first place helps reinforce the value of this baseline work. The dynamics are explored in the guesswork gap: using specification-true quotes to build a predictable folding carton packaging budget.
Audit Completion Verification
Use this 10-point self-audit checklist to verify the audit is complete:
1. All current folding carton specification files exist in one working audit set. Hidden omissions usually sit across multiple files rather than inside one obvious error. Scattered files hide contradictions that only surface when suppliers interpret them differently.
2. State the active board grade explicitly for every relevant SKU or family. Unstated material assumptions are where quote variance begins—a dynamic explored in the guesswork gap: using specification-true quotes to build a predictable folding carton packaging budget.
3. Barrier-property requirements are explicitly stated rather than assumed. Pharmaceutical products often require specific moisture, oxygen, or light barriers that vary by product and storage condition. Vague protective requirements often mask significant technical discrepancies between vendor capabilities.
4. Coatings or substrate treatments are named where applicable. Unstated coating expectations create post-production disputes that specifications should have prevented. For guidance on defining frozen-food barriers, which share similar coating considerations with some pharmaceutical applications, see how to define safe contact barriers for frozen food folding carton packaging. Surface choices affect both protection and print behavior.
5. Print-critical tolerance expectations are documented clearly enough for supplier interpretation. Vague print language invites supplier guesswork on registration, contrast, and line weight. An undefined quality standard is not a production instruction.
6. Current files state requirements that support 2D matrix readability rather than generic barcode language. Serialization mandates require specific DataMatrix performance that generic specifications cannot ensure. Readability problems often start at the specification stage.
7. Test methods or verification expectations are named where practical and relevant. Downstream scan verification depends on upstream print quality that specifications should define. For guidance on structuring proof requirements, see what proof to request at RFQ stage for folding cartons. An undefined check is hard to enforce consistently.
8. No supplier-provided data sheets are being treated as the house specification. External documents transfer baseline control outside the organization. This risk is examined in detail in the danger of vendor-led data sheets: establishing rigid folding carton specification requirements. Buyer control weakens when supplier documents become the baseline.
9. Shared requirements are separated from SKU-specific exceptions. Mixed documentation creates confusion across product families and complicates supplier communication. Structural guidance for managing multiple SKUs appears in the specification bridge: a practical blueprint for folding carton requirements across multiple SKUs. Mixed logic creates avoidable over-application.
10. Every missing or vague field has been captured in an updated baseline blueprint. The audit creates value only if gaps are formally addressed in a consolidated document. An audit only matters when the findings become the new reference point.
What To Do After The Audit
A completed audit produces a specification-true baseline. The next step is applying that baseline to supplier evaluation—ensuring that quotes reflect the same underlying assumptions and can be compared on equal terms. Teams ready to begin supplier outreach can find suppliers through PaperIndex’s global directory.
For practical guidance on quote normalization and the next steps in specification-driven sourcing, continue with how to normalize your first folding carton quote: a beginner’s guide.
If the biggest gaps are material-related, board grade tolerances explained: securing folding carton specifications across suppliers is the most direct follow-on. If the audit exposed too much dependence on vendor paperwork, the truth decay of data sheets: why relying on supplier specifications ruins folding carton specifications reinforces the case for a buyer-owned baseline.
For more methodology-first packaging guidance, explore the PaperIndex Academy.
Disclaimer:
This article provides general educational guidance on pharmaceutical packaging specification audits. Regulatory requirements, including DSCSA and EU FMD frameworks, are subject to change; verify current obligations with official sources. Specific tolerances, test methods, and verification standards should be determined in consultation with qualified packaging engineers, quality professionals, and regulatory experts based on your products and markets.
Our Editorial Process:
Our expert team uses AI tools to help organize and structure our initial drafts. Every piece is then extensively rewritten, fact-checked, and enriched with first-hand insights and experiences by expert humans on our Insights Team to ensure accuracy and clarity.
About the PaperIndex Insights Team:
The PaperIndex Insights Team is our dedicated engine for synthesizing complex topics into clear, helpful guides. While our content is thoroughly reviewed for clarity and accuracy, it is for informational purposes and should not replace professional advice.

